
C57BL/6-Tfrctm1(TFRC)Bcgen/Bcgen • 110861
Background:
Validation:
Application:
Gene targeting strategy for B-hTFR1 mice. The exons 4-19 of mouse Tfr1 gene that encode extracellular domain are replaced by human counterparts in B-hTFR1 mice. The genomic region of mouse Tfr1 gene that encodes cytoplasmic portion is retained. The promoter, 5’UTR and 3’UTR region of the mouse gene are also retained. The chimeric TFR1 expression is driven by endogenous mouse Tfr1 promoter, while mouse Tfr1 gene transcription and translation will be disrupted.
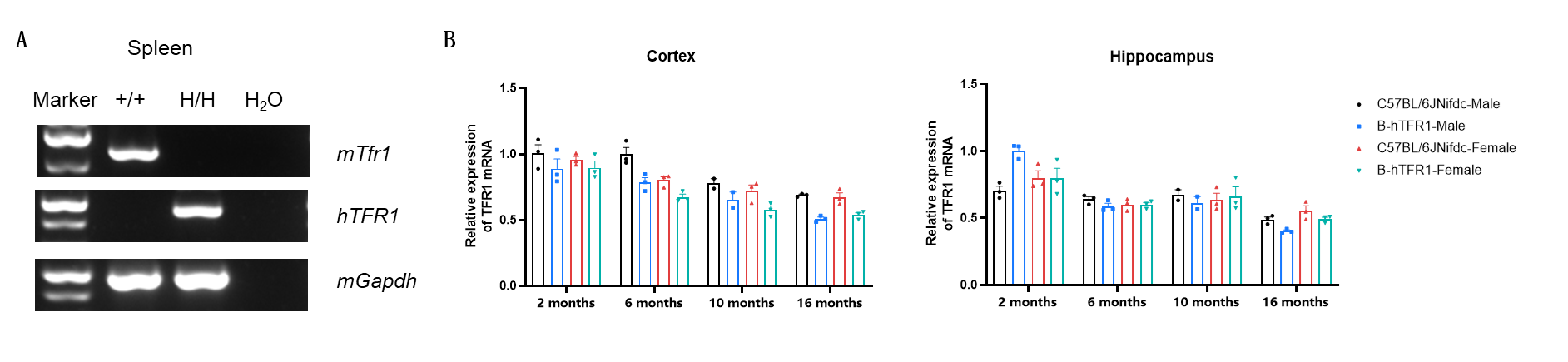
Strain-specific TFR1 expression analysis in wild-type C57BL/6 mice and homozygous B-hTFR1 mice. (A) Spleen RNA were isolated from wild-type C57BL/6 mice (+/+) and homozygous B-hTFR1 mice (H/H), then cDNA libraries were synthesized by reverse transcription, followed by PCR with mouse or human TFR1 primers. (B) Quantitative real-time PCR (qRT-PCR) analysis of relative TFR1 mRNA expression in the cortex (left) and hippocampus (right) of male and female wild-type C57BL/6JNifdc mice and homozygous B-hTFR1 mice at 2, 6, 10, and 16 months of age. Expression levels were normalized to TFR1 expression in cortex of 2-month-old C57BL/6JNifdc male mice. Values are expressed as mean ± SEM.
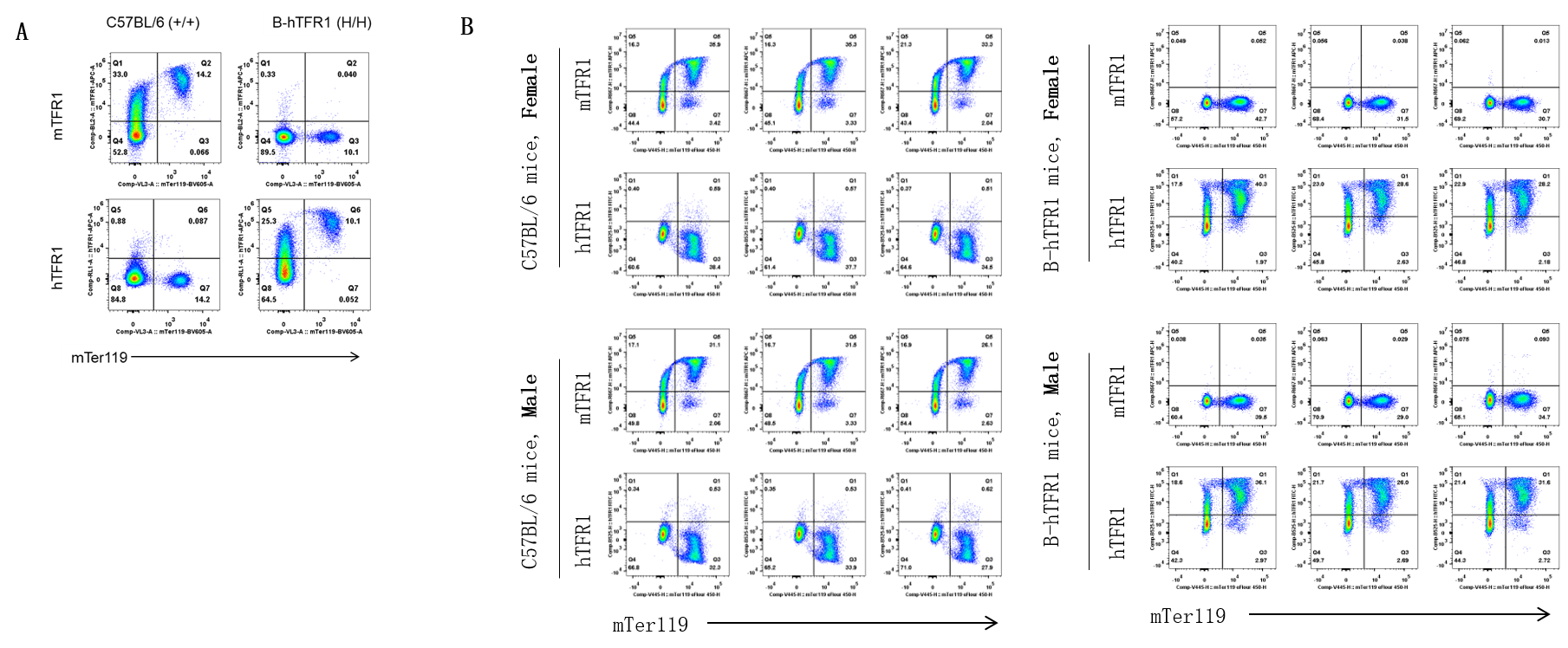
Strain-specific TFR1 expression analysis in wild-type C57BL/6 mice and homozygous B-hTFR1 mice. Bone marrow erythrocytes were collected from wild-type C57BL/6 mice (+/+) and homozygous B-hTFR1 mice (H/H). Protein expression was analyzed with anti-mouse TFR1 antibody (Biolegend, 113807) and anti-human TFR1 antibody (Biolegend, 334107) by flow cytometry.
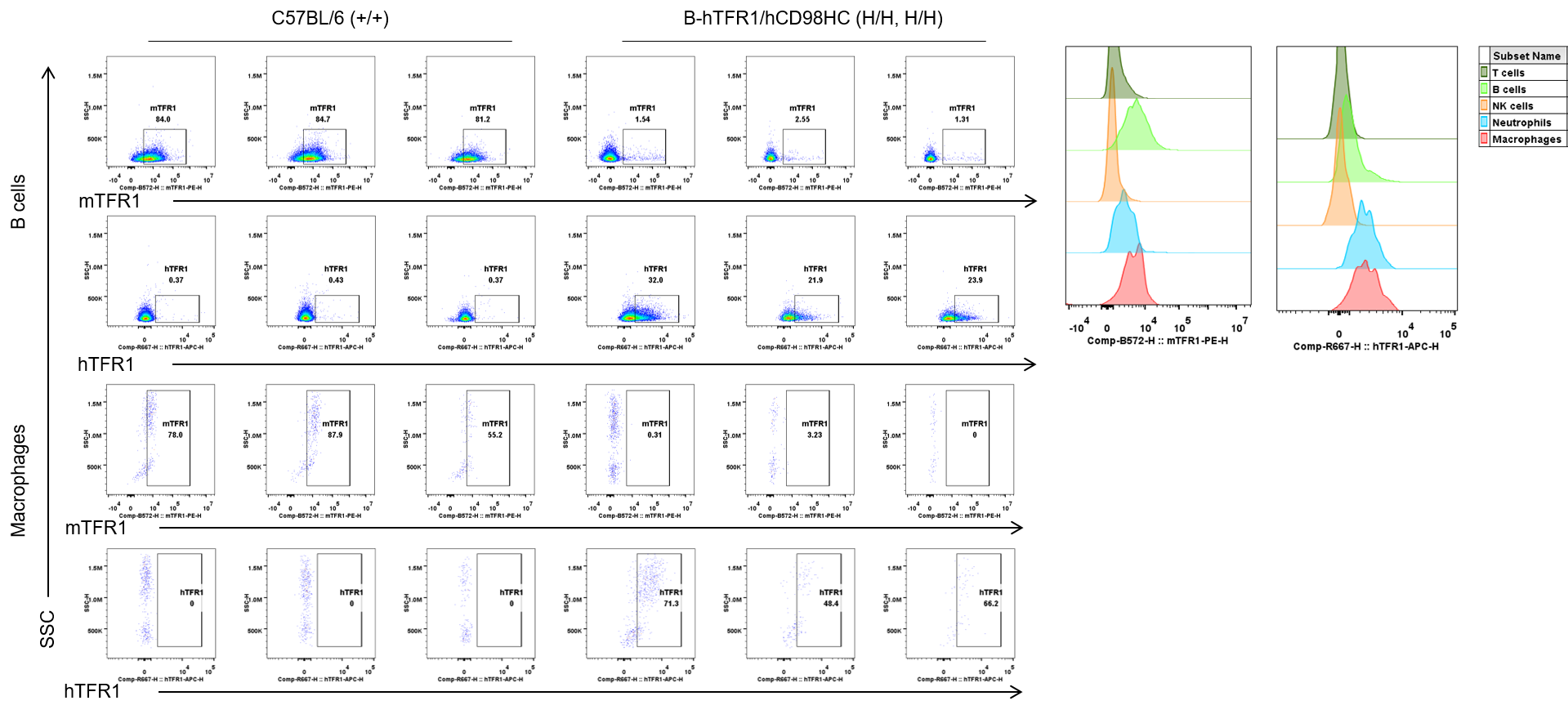
Strain specific TFR1 expression analysis in wild-type C57BL/6 and homozygous B-hTFR1/hCD98HC mice by flow cytometry. Blood cells were collected from wild-type C57BL/6 (+/+) and homozygous B-hTFR1/hCD98HC mice (H/H, H/H) and analyzed by flow cytometry with anti-mouse TFR1 antibody (Biolegend, 113808) and anti-human TFR1 antibody (Biolegend, 334108). mTFR1 was only detectable in wild-type mice, and hTFR1 was exclusively detectable in B cells and macrophages from homozygous mice but not from wild-type mice.
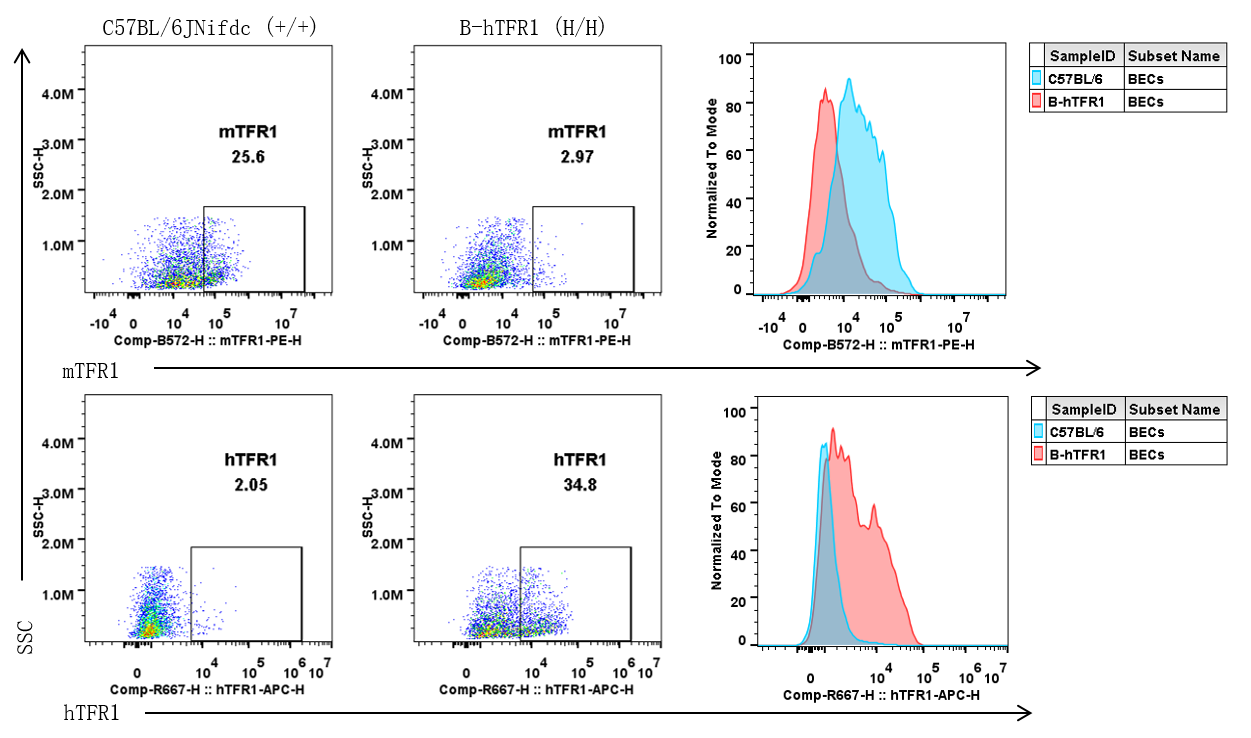
Strain specific TFR1 expression analysis in wild-type C57BL/6JNifdc and homozygous B-hTFR1 mice by flow cytometry. Brain cells were collected from wild-type C57BL/6JNifdc (+/+) and homozygous B-hTFR1 mice (H/H), and analyzed by flow cytometry with anti-mouse TFR1 antibody (Biolegend, 113808) and anti-human TFR1 antibody (Biolegend, 334108).
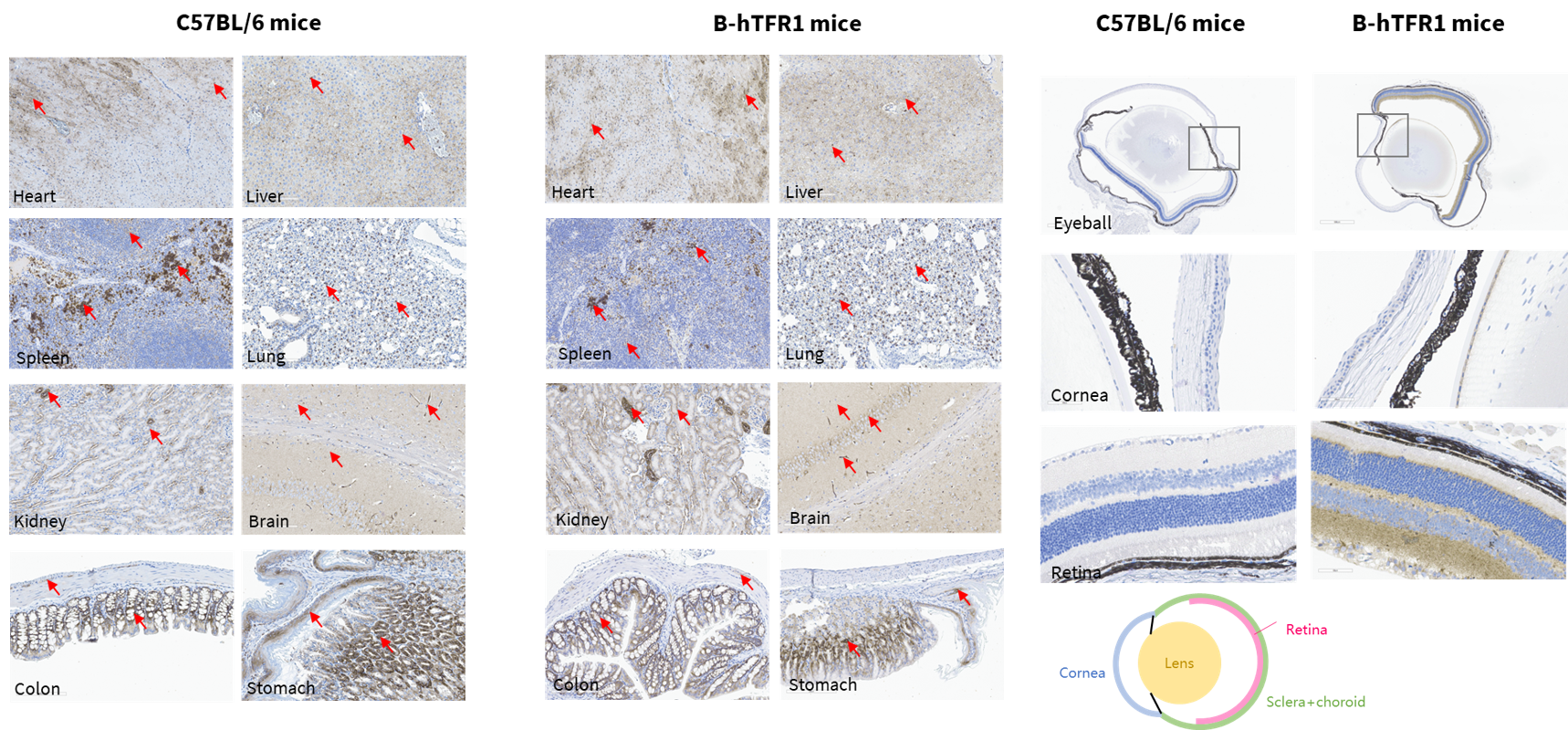
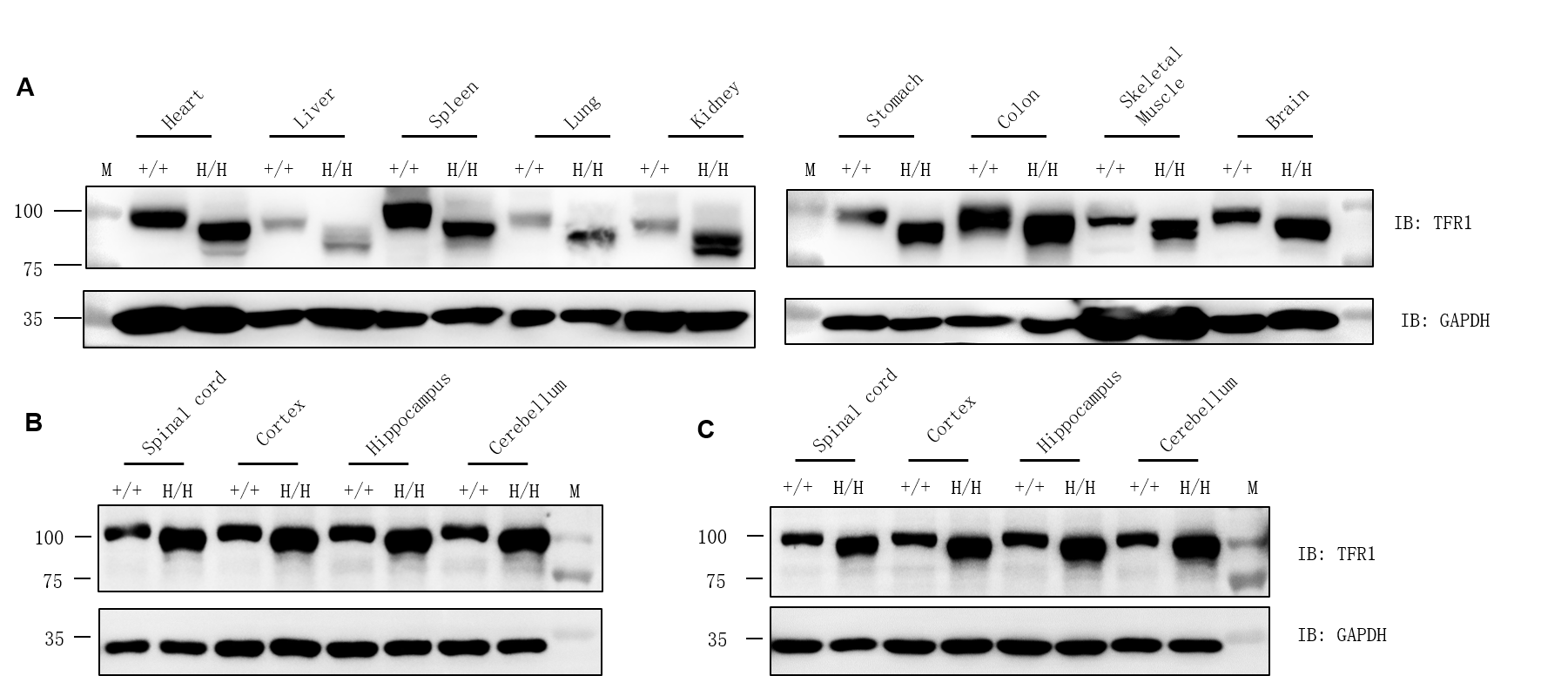
TFR1 protein expression analysis by western blotting using a cross-reactive anti-TFR1 antibody (abcam, ab214039). M, marker. (B) Male. (C) Female.
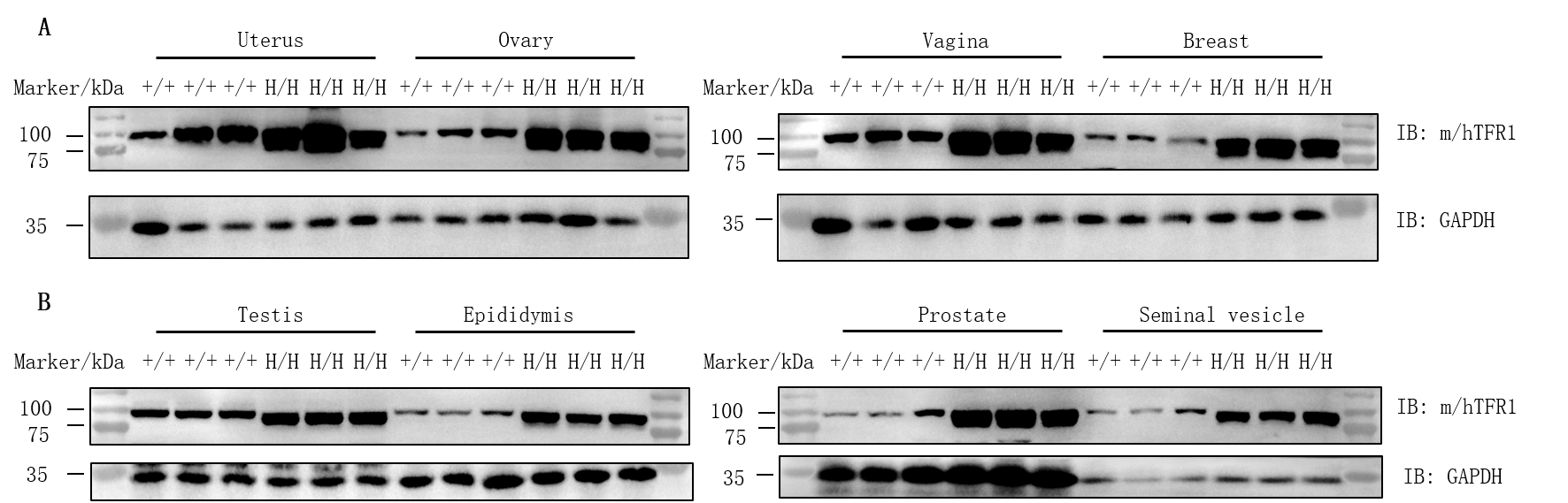
Protein expression analysis of TFR1 in homozygous B-hTFR1/hCD98HC mice. Various tissue lysates were collected from wild-type C57BL/6JNifdc mice (+/+) and homozygous B-hTFR1/hCD98HC mice (H/H), and then analyzed by western blot with anti-transferrin receptor antibody (abcam, ab214039) and anti-CD98 antibody (abcam, ab307587). 30 μg total proteins were loaded for western blotting analysis. TFR1 was detected in uterus, ovary, vagina, breast, testis, epididymis, prostate and seminal vesicle from both wild-type C57BL/6JNifdc mice and homozygous B-hTFR1/hCD98HC mice, as the antibody was cross-reactive between human and mouse.
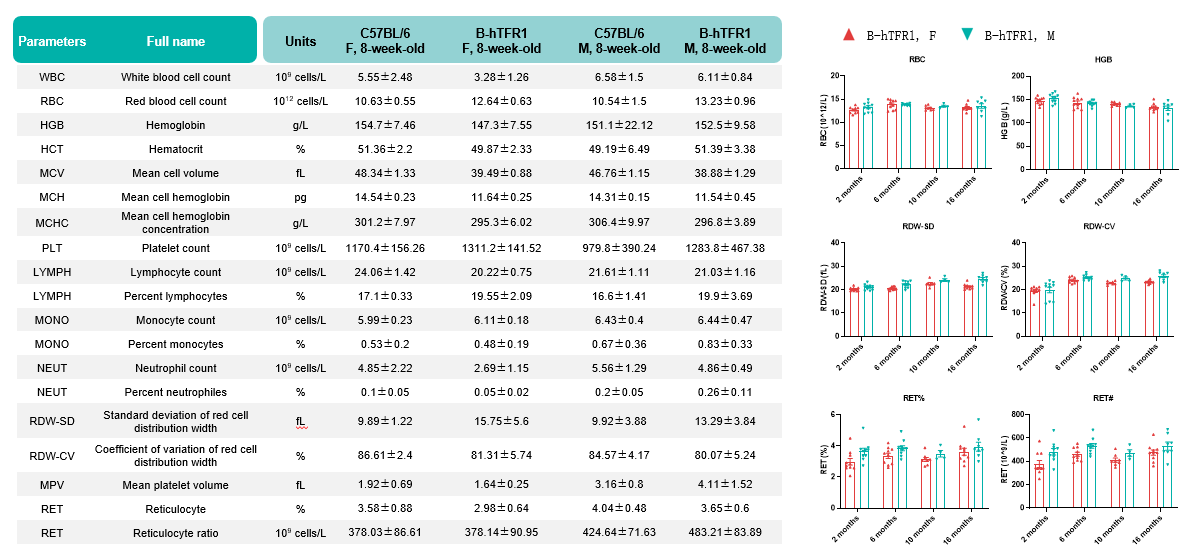
Values are expressed as mean ± SD.
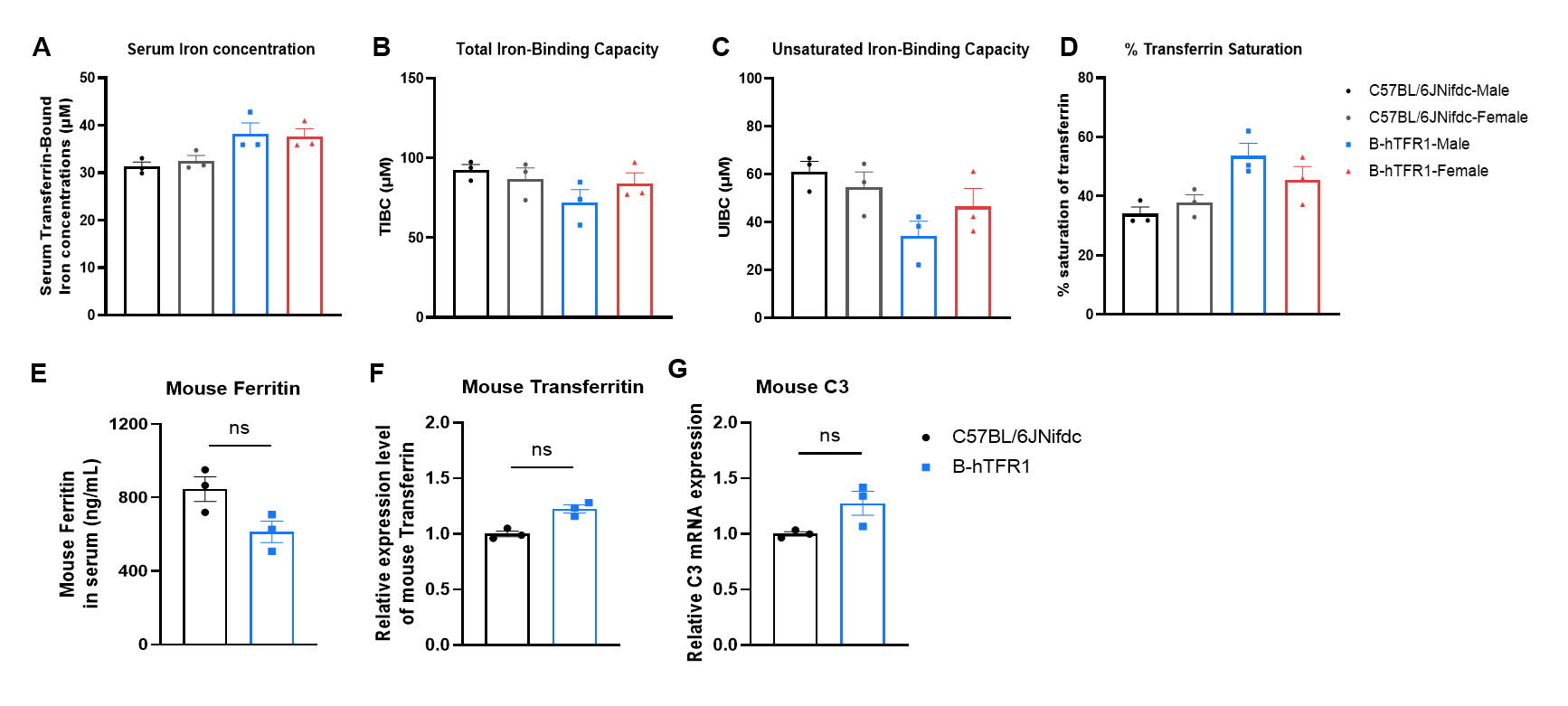
Serum iron homeostasis assay in wild-type C57BL/6JNifdc and homozygous humanized B-hTFR1 mice. (A-D) Serum were collected from C57BL/6JNifdc (female, n=3; male, n=3; 14-week-old) and B-hTFR1 mice (female, n=3; male, n=3; 9-week-old), and then analyzed by Total Iron-Binding Capacity (TIBC) and Serum Iron Assay kit (abcam, ab239715). (E-G) Mouse ferritin, transferritin and complement 3 detection in wild-type C57BL/6JNifdc and B-hTFR1 mice (male, n=3, 8-week-old). Values are expressed as mean ± SEM. Significance was determined by two-way ANOVA test. *P < 0.05, **P < 0.01, ***p < 0.0001.
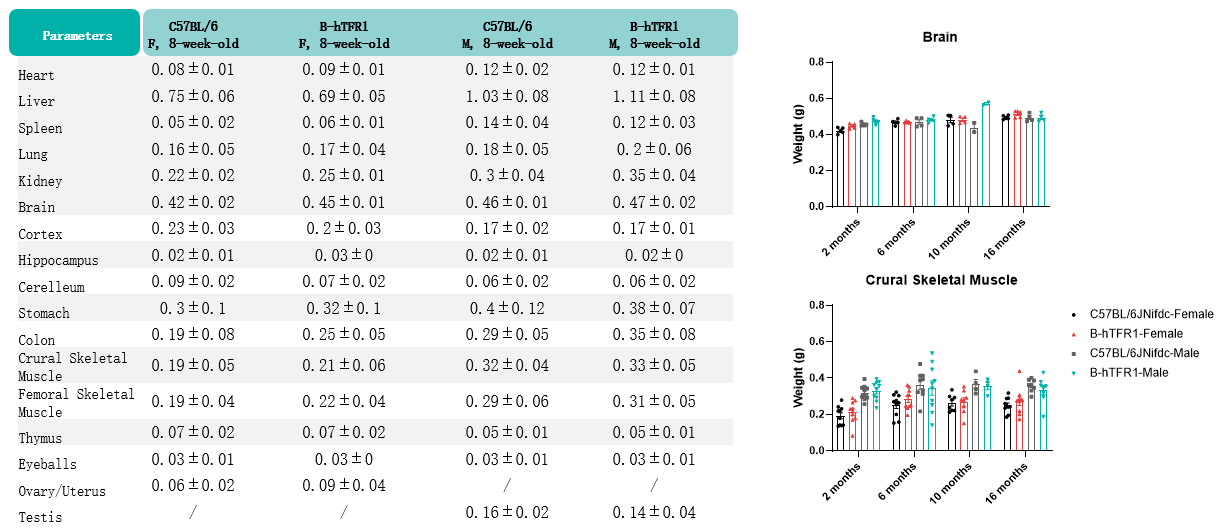
Values are expressed as mean ± SD.
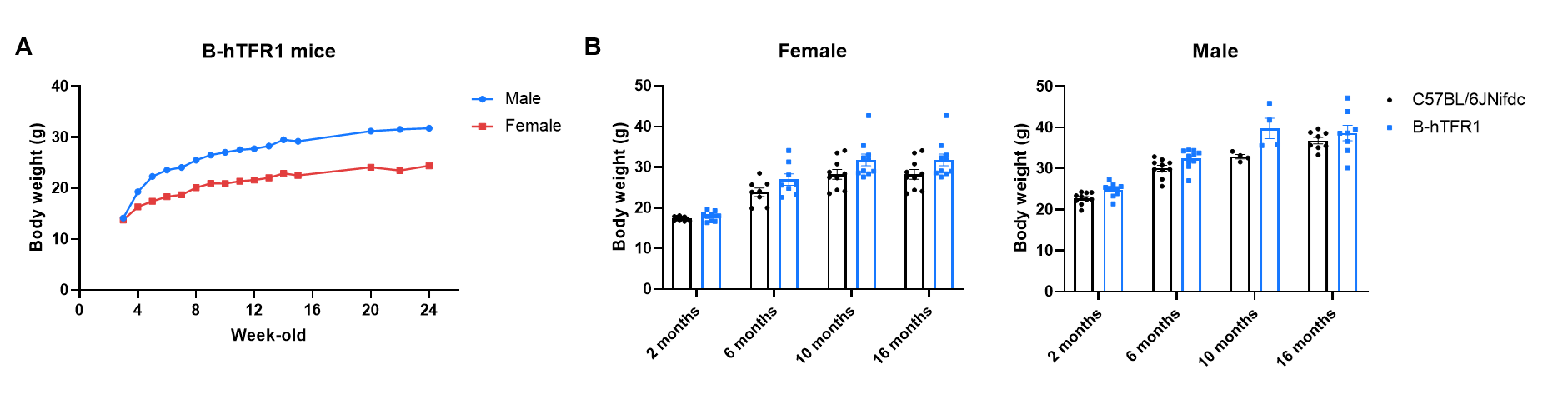
Body weight growth of B-hTFR1 mice compared with C57BL/6JNifdc mice. (A) Longitudinal body weight of male and female B-hTFR1 mice from 3 to 24 weeks of age. (B) Body weight of female and male B-hTFR1 mice at 2, 6, 10, and 16 months of age, compared with age- and sex-matched wild-type C57BL/6JNifdc mice (black bars). Values are expressed as mean ± SEM.
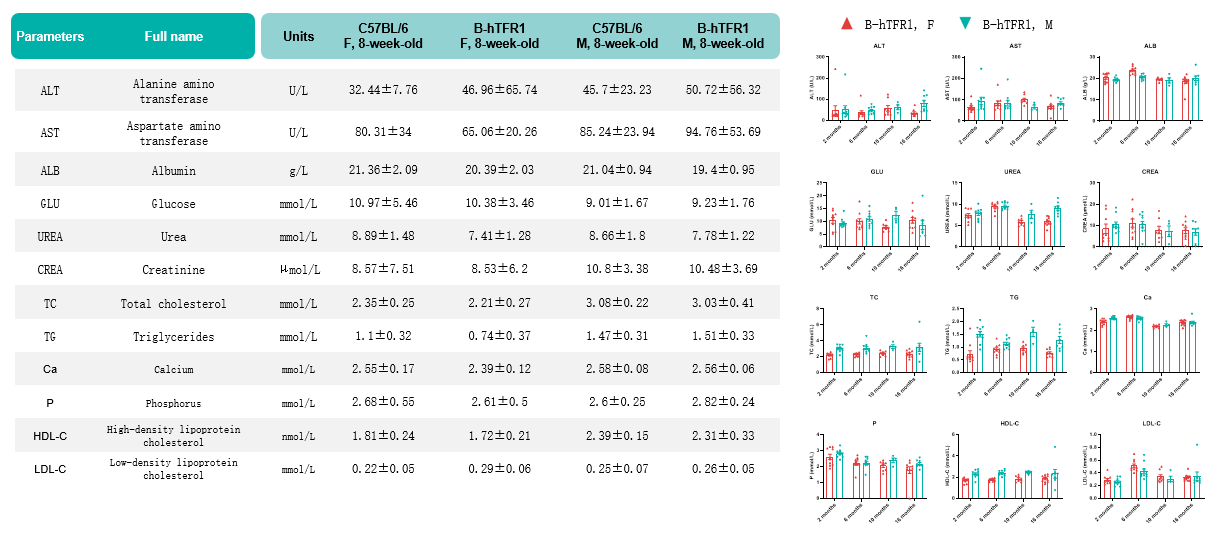
Values are expressed as mean ± SD.
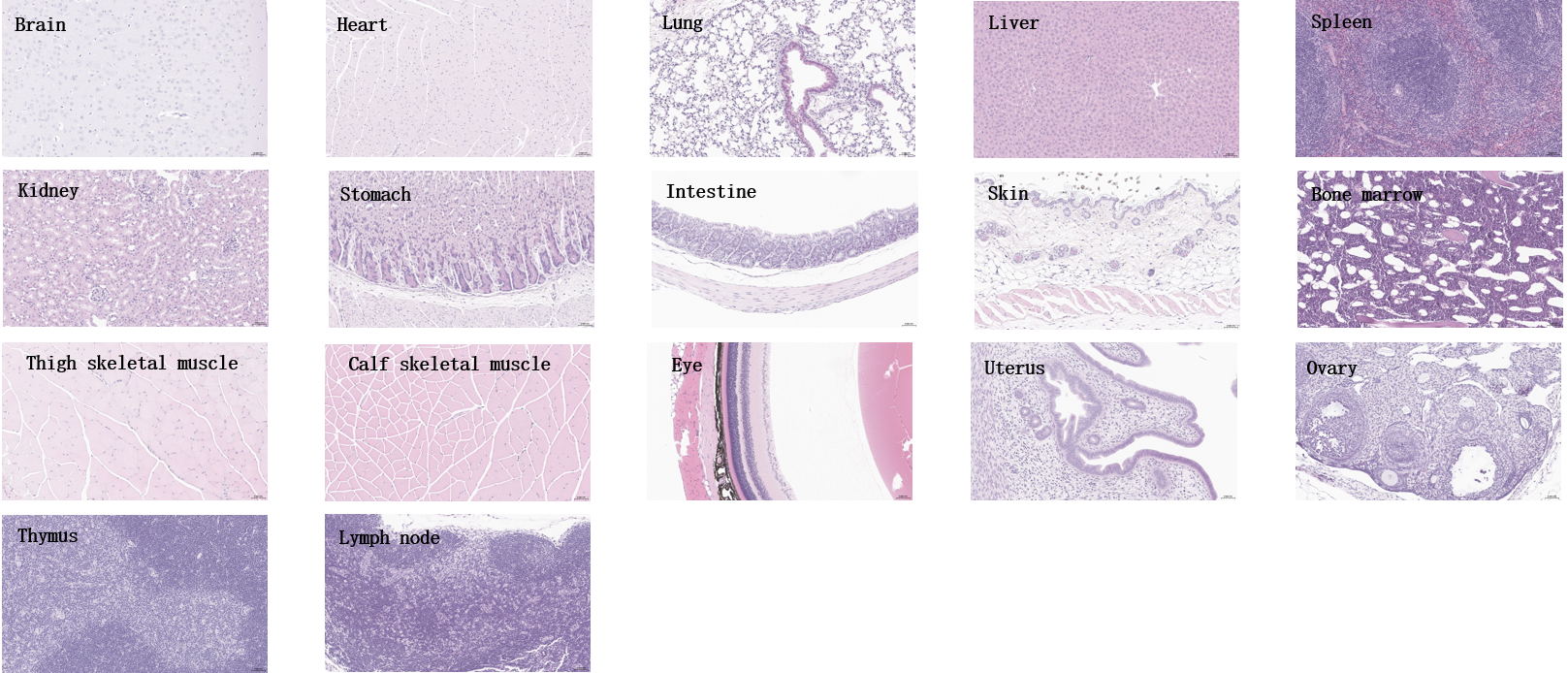
Histopathological analysis of major organs from homozygous B-hTFR1 mice. Tissues were collected from B-hTFR1 mice (female, 8-week-old, n=3), fixed in 4% paraformaldehyde and stained with hematoxylin and eosin (H&E). Representative photomicrographs are shown. Scale bar: 40x.
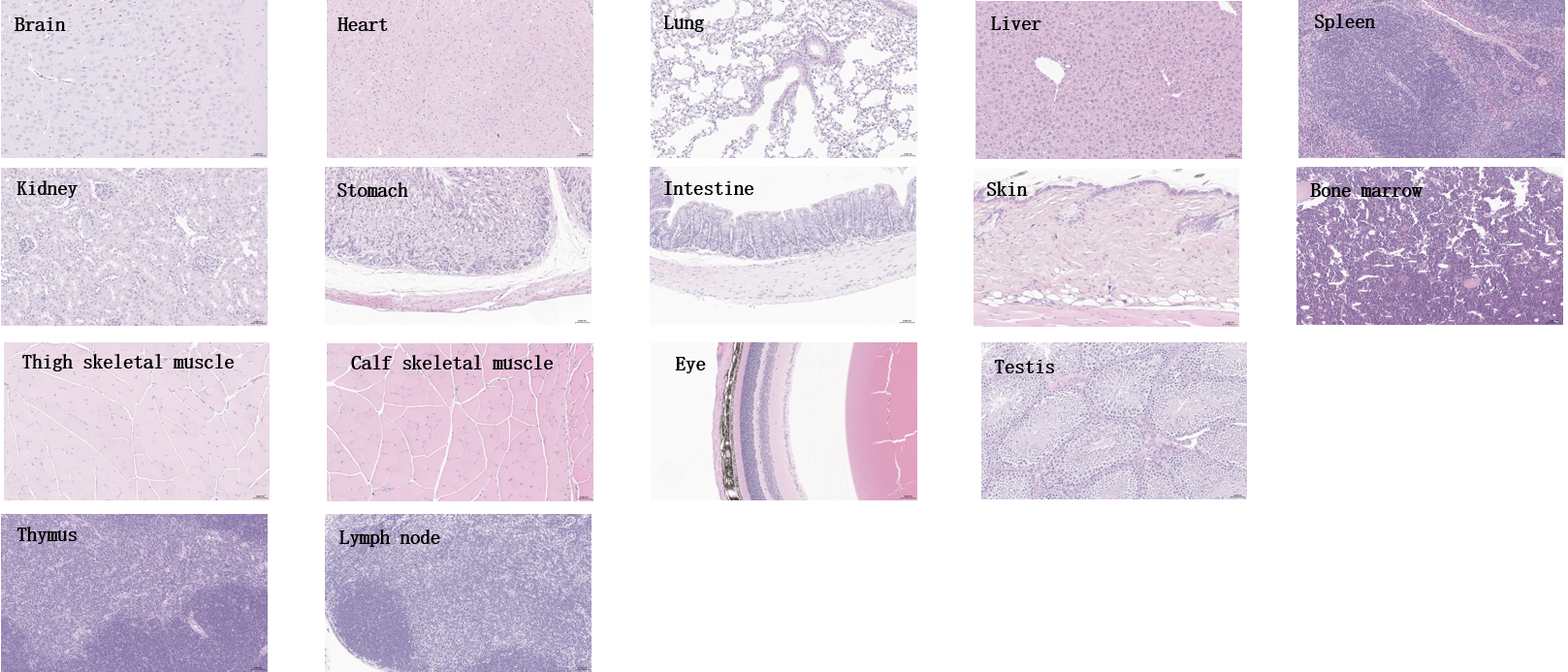
Histopathological analysis of major organs from homozygous B-hTFR1 mice. Tissues were collected from B-hTFR1 mice (male, 8-week-old, n=3), fixed in 4% paraformaldehyde and stained with hematoxylin and eosin (H&E). Representative photomicrographs are shown. Scale bar: 40x.
Values are expressed as mean ± SD.

Body composition analysis of wild-type C57BL/6JNifdc mice and homozygous B-hTFR1 mice (6-month-old, n=10) measured by Body Composition Analyzer. (A) Absolute body weight, lean mass, fat mass, and free water mass (expressed in grams). (B) Relative lean content, fat content, free water content, total content, and total water content (expressed as percentages). B-hTFR1 mice exhibited normal body composition profiles with no notable differences from wild-type C57BL/6JNifdc mice.
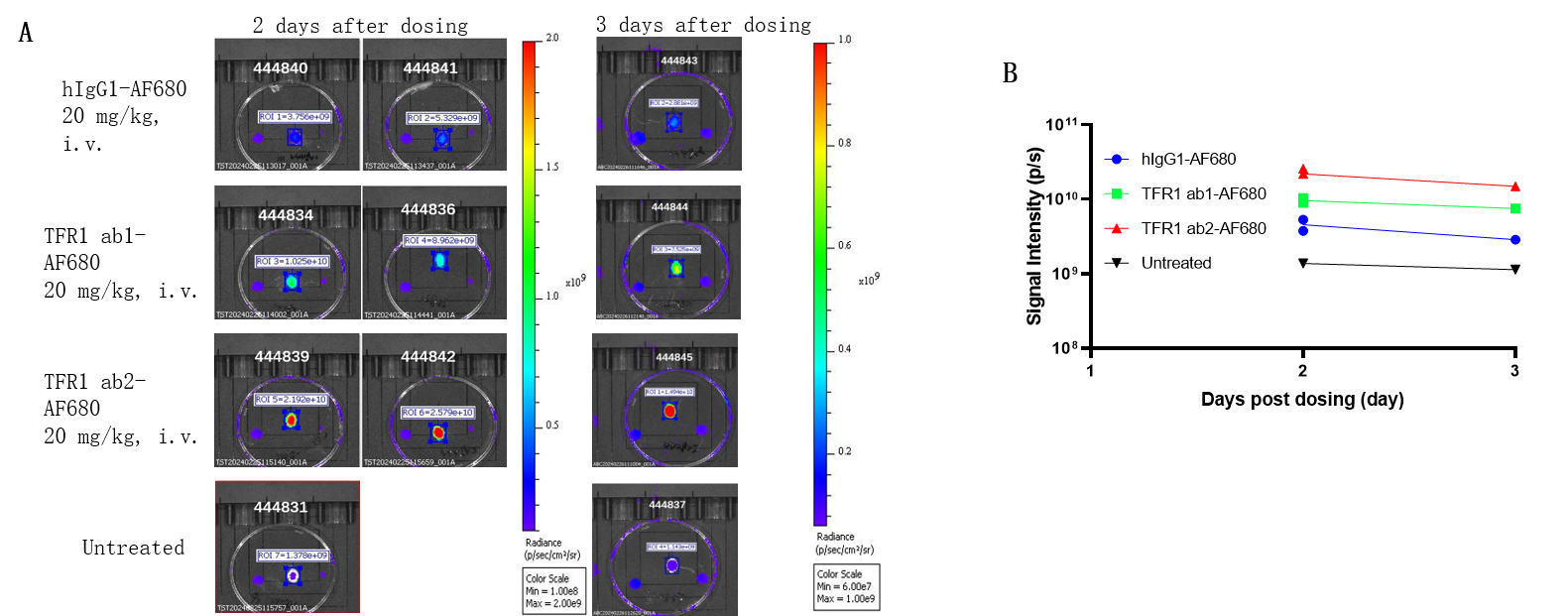
B-hTFR1 mice were intravenously injected with AF680-conjugated control hIgG1 or anti human TFR1 antibodies ab1 and ab2 (provided by a client). After 2 days or 3 days post-injection, the mice were perfused and their brains were collected for analysis. (A) Mouse brain images under imaging system. (B) Fluorescence intensity of mouse brain under imaging system. The results indicate that the uptake of anti-human TFR1 antibody ab2 in the brain of B-hTFR1 mice was higher than that of anti-human TFR1 antibody ab1.
Note: These data were obtained from collaborative validation with the client, who provided the antibody.
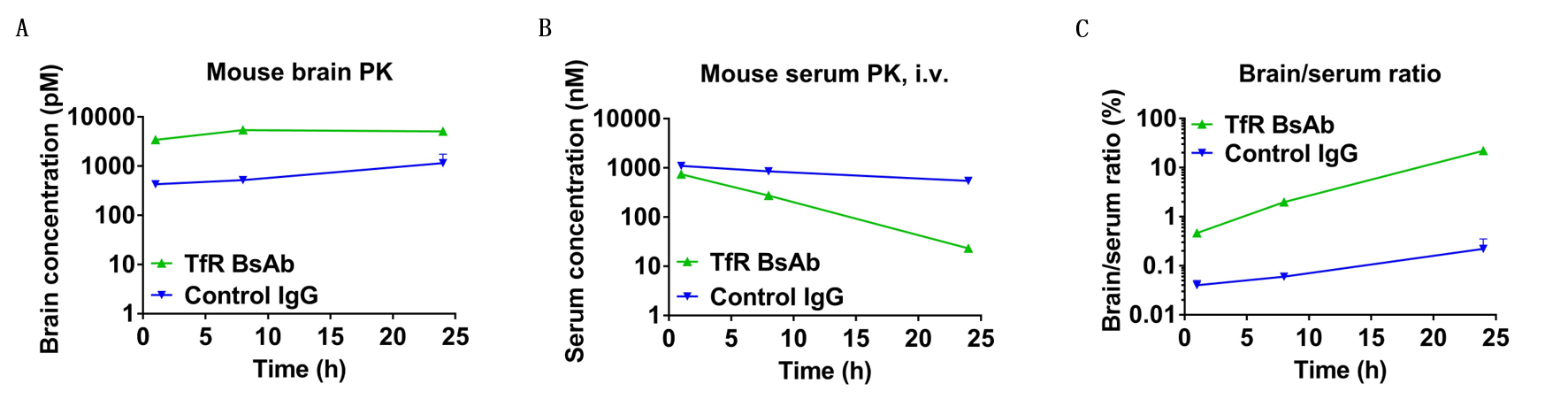
In vivo pharmacokinetic (PK) evaluation of anti-human TFR1 bispecific antibodies (BsAbs). B-hTFR1 mice were injected with control IgG (10 mpk) and anti-human TFR1 BsAbs (10.9 mpk) provided by a client via tail vein. Brain and serum were taken for in vivo PK evaluation. Brain concentrations(A), serum concentrations (B), and brain-to-serum ratio (C) of anti-human TFR1 BsAbs were quantified. As shown in panel, anti-human TFR1 BsAbs exhibited higher serum clearance and enhanced brain exposure after dose. The results confirmed that brain of B-hTFR1 mice enables uptake of an intravenously administered anti-human TFR1 BsAbs and B-hTFR1 mice provide a powerful preclinical model for in vivo evaluation of effective delivery of protein therapeutics to the central nervous system (CNS). Graphs represent mean ± SEM.
Note: This experiment was performed by the client using B-hTFR1 mice. All the other materials were provided by the client.
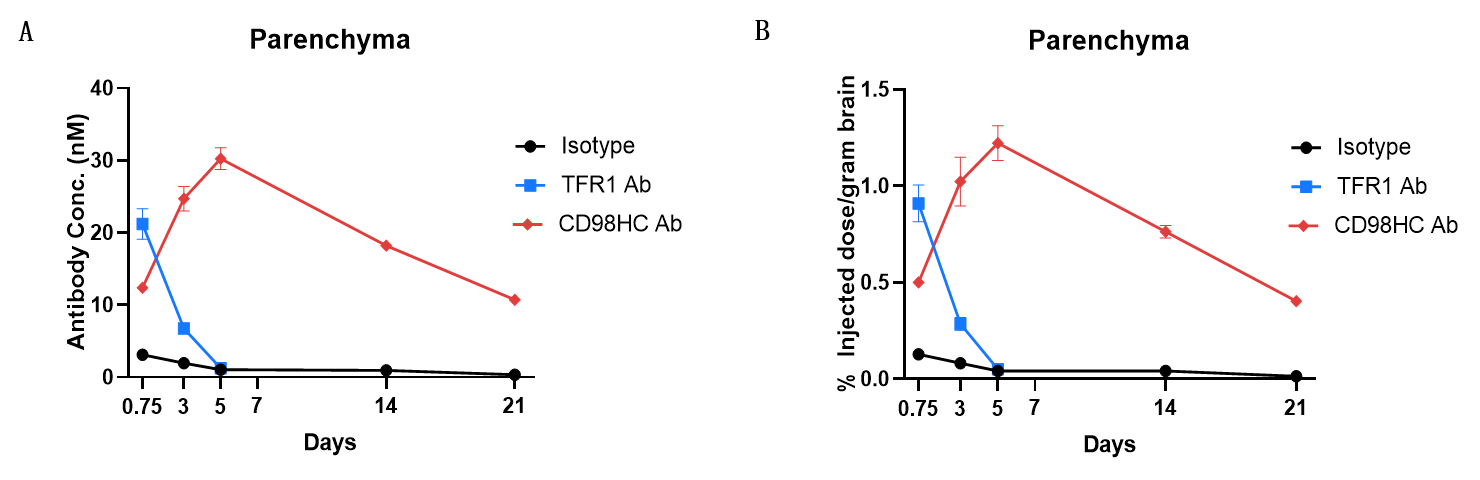
In vivo PK evaluation and comparison of anti-human TFR1 and anti-CD98HC antibody. B-hTFR1/hCD98HC mice (n=2, female, 8-week-old) were injected with control IgG (10 mpk) anti-human TFR1 antibody (TFR1 Ab, JR-141 analog, monovalent, produced in house, 12.56 mpk) and anti-human CD98HC antibody (CD98HC Ab, CD98BBBB-h1.L analog, monovalent, produced in house, 13.3 mpk) via tail vein. Brain were taken for in vivo PK evaluation after dosing 18 h and 3, 5, 14, 21 days. Brain concentrations (A) and % of injection/gram brain (B) were quantified. As shown in panel, anti-human TFR1 antibody exhibited higher brain exposure in 24 h after dose, while anti-CD98HC antibody exhibited higher brain exposure in 72 h after dose. The results confirmed that B-hTFR1/hCD98HC mice enables uptake of an intravenously administered anti-human TFR1 antibody or anti-human CD98HC antibody, and this mice can be used for the comparison of penetration efficacy of shuttle molecules targeting TFR1 or CD98HC. Graphs represent mean ± SEM.
B-hTFR1/hTAU Mice: Gene Targeting Strategy
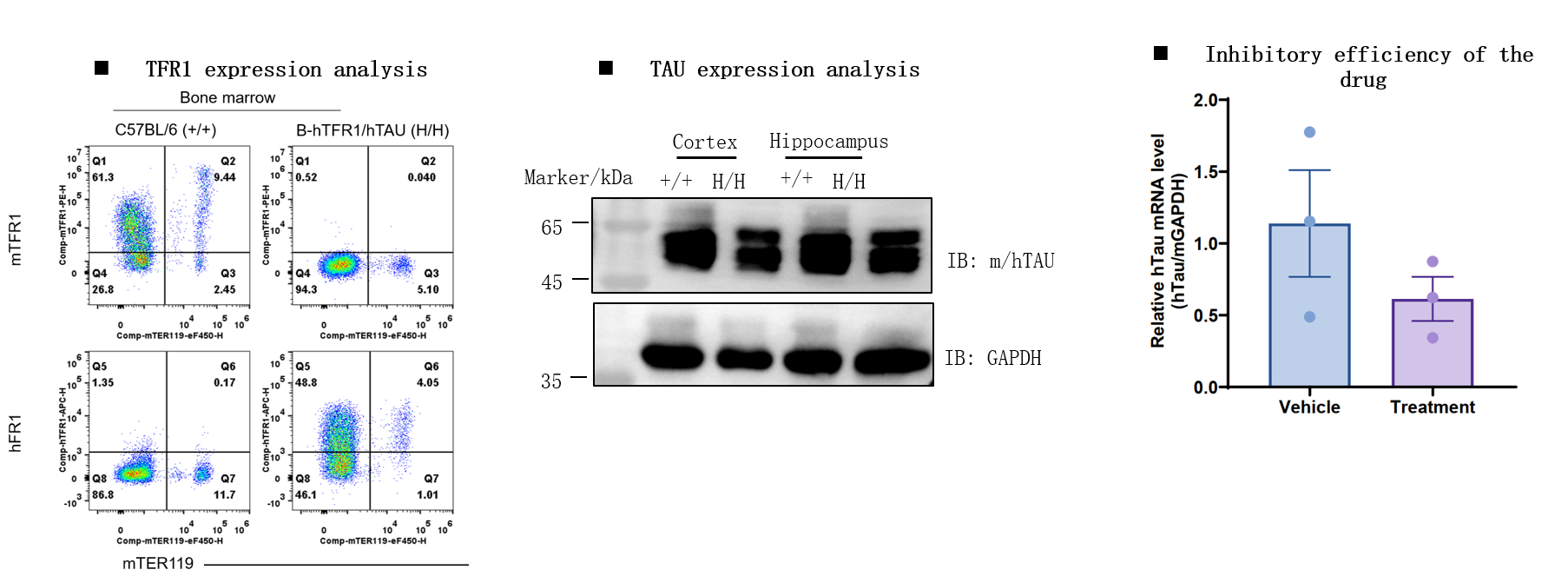
Note: This experiment was performed by the client using B-hTFR1/hTAU mice. All the other materials were provided by the client (The detailed information regarding the drug, administration strategy, dosage, and timing are un-disclosed).
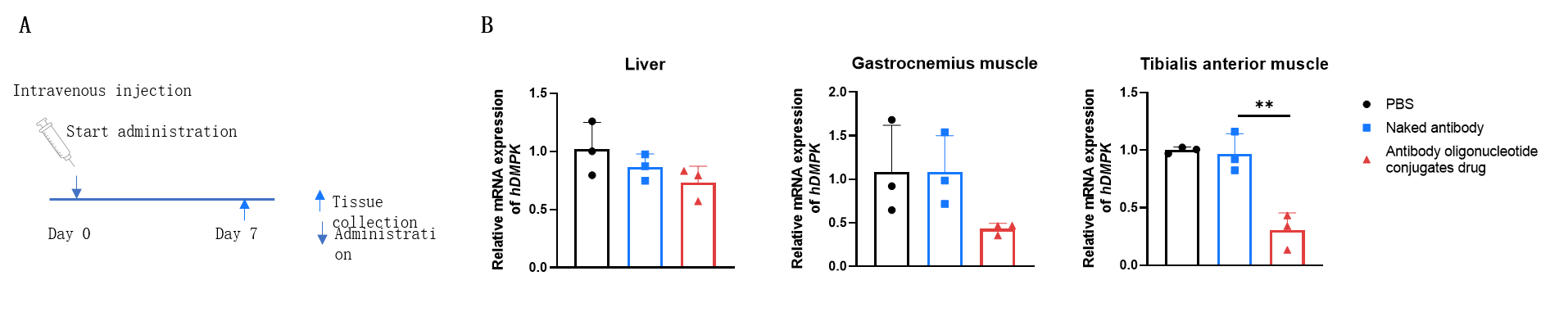
The inhibitory efficiency of the antibody oligonucleotide conjugates drug against human DMPK in heterozygous B-hTFR1/hDMPK mice. The antibody oligonucleotide conjugates drug (3 mpk, produced in-house), naked antibody (3 mpk, produced in-house) and PBS were administered to the heterozygous B-hTFR1/hDMPK mice individually on day 0. The mice were sacrificed on day 7, and the liver, gastrocnemius muscle and tibialis anterior muscle were collected to detect the expression level of human DMPK mRNA by qPCR. The human DMPK mRNA in the treatment groups (antibody oligonucleotide conjugates drug) were significantly reduced compared to the control groups (naked antibody and PBS) in tibialis anterior muscle, demonstrating that B-hTFR1/hDMPK mice provide a powerful preclinical model for in vivo evaluation of human DMPK targeted antibody oligonucleotide conjugates drug. Values are expressed as mean ± SEM.
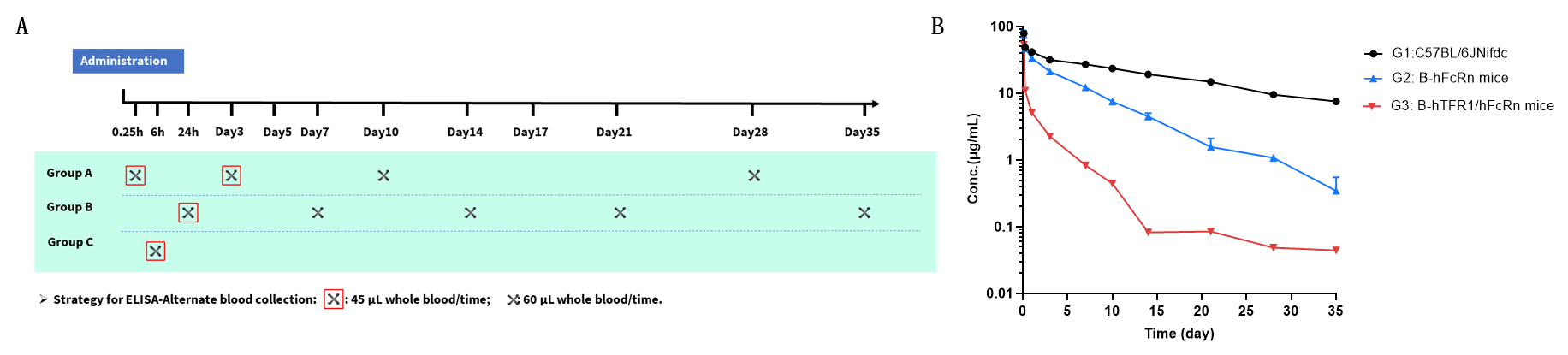
Pharmacokinetic (PK) analysis of JR-141 analog in wild-type and humanized TFR1/FcRn mouse strains. (A) Scheme design. Male mice (n = 9 per group) from wild-type C57BL/6 mice (G1), B-hFcRn mice (G2) and B-hTFR1/hFcRn mice (G3) received a single intravenous (i.v.) dose of 3 mg/kg JR-141 analog. Blood samples were collected at 0.25 h, 6 h, 24 h, and on days 3, 7, 10, 14, 21, 28, and 35 post-administration. (B) Serum concentrations of JR-141 analog were measured by ELISA. Compared to wild-type mice (G1), B-hFcRn mice (G2) and B-hTFR1/hFcRn mice (G3) exhibited accelerated clearance. B-hTFR1/hFcRn mice better reflect in vivo antibody concentration changes compared to wild-type C57BL/6JNifdc and B-hFcRn mice, especially valuable for evaluating the PK of antibodies exhibiting target-mediated drug disposition (TMDD) in a more physiologically relevant context.
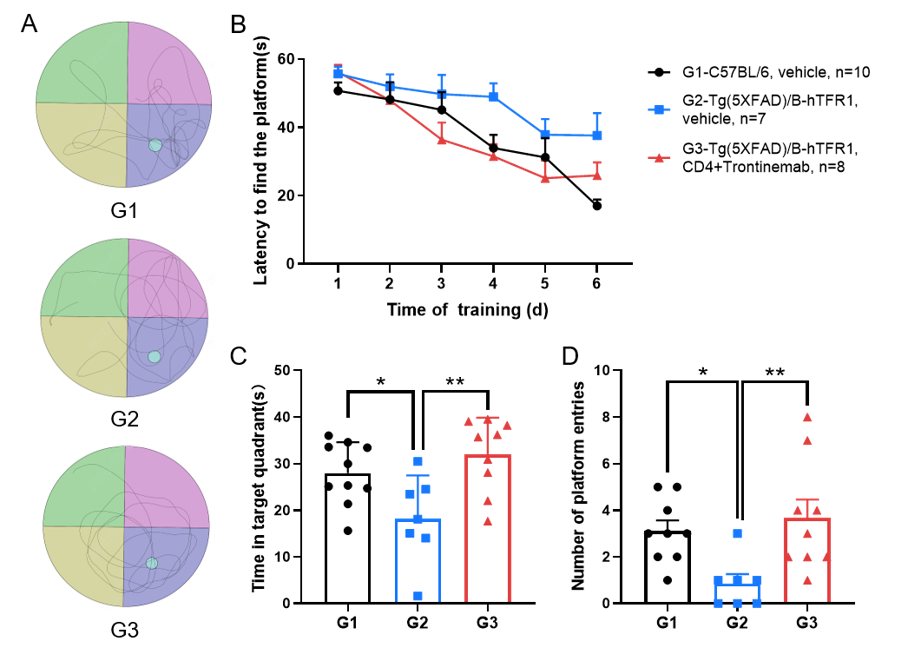
This experiment was conducted in collaboration with the client using Tg(5XFAD)/B-hTFR1 mice.