• 322200

| Product name | B-hHER2 CT26.WT |
|---|---|
| Catalog number | 322200 |
| Strain background | BALB/c |
| NCBI gene ID | 13866 (Human) |
| Aliases | Neu; HER2; HER-2; c-neu; Erbb-2; c-erbB2; l11Jus8; mKIAA3023 |
| Tissue | Colon |
| Disease | Colon carcinoma |
| Species | Mouse |
| Application | B-hHER2 CT26.WT cells have the capability to establish tumors in vivo |
Gene targeting strategy for B-hHER2 CT26.WT cells. The exogenous promoter and human HER2 coding sequence was inserted to replace part of murine exon 2 and all of exons 3-7. The insertion disrupts the endogenous murine Her2 gene, resulting in a non-functional transcript.
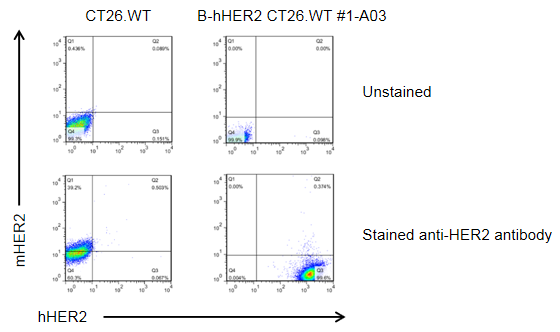
HER2 expression analysis in B-hHER2 CT26.WT cells by flow cytometry. Single cell suspensions from wild-type MC38 and B-hHER2 CT26.WT #1-A03 cultures were analyzed with anti-mouse HER2 antibody (RD, FAB6744P) and anti-human HER2 antibody (Biolegend, 324408) by flow cytometry. Human HER2 was detected on the surface of B-hHER2 CT26.WT cells but not wild-type MC38 cells.
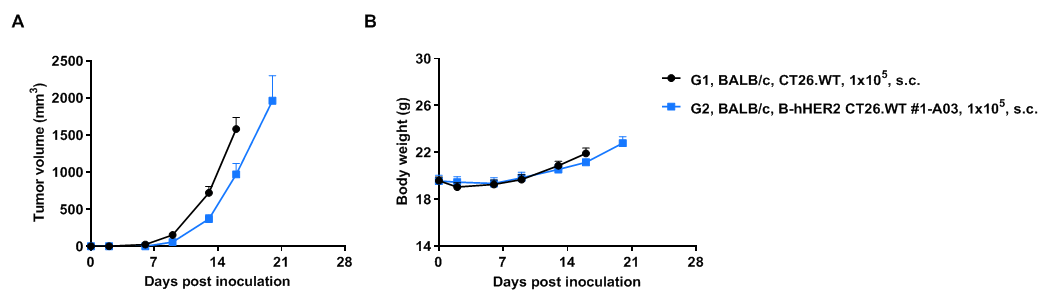
Subcutaneous tumor growth of B-hHER2 CT26.WT cells. B-hHER2 CT26.WT cells (1x105) and wild-type MC38 cells (1x105) were subcutaneously implanted into BALB/c mice (female, 6-7-week-old, n=6). Tumor volume and body weight were measured twice a week. (A) Average tumor volume. (B) Body weight. Volume was expressed in mm3 using the formula: V=0.5 X long diameter X short diameter2. Results indicate that B-hHER2 CT26.WT cells were able to establish tumors in vivo and can be used for efficacy studies. Values are expressed as mean ± SEM.
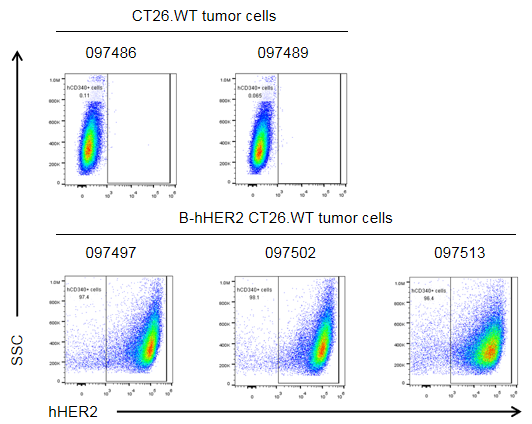
HER2 expression evaluated on B-hHER2 CT26.WT tumor cells by flow cytometry. B-hHER2 CT26.WT cells were subcutaneously transplanted into BALB/c mice (n=6). Upon conclusion of the experiment, tumor cells were harvested and analyzed with anti-human HER2 antibody (Biolegend, 324406) by flow cytometry. As shown, human HER2 was highly expressed on the surface of tumor cells. Therefore, B-hHER2 CT26.WT cells can be used for in vivo efficacy studies evaluating novel HER2 therapeutics.
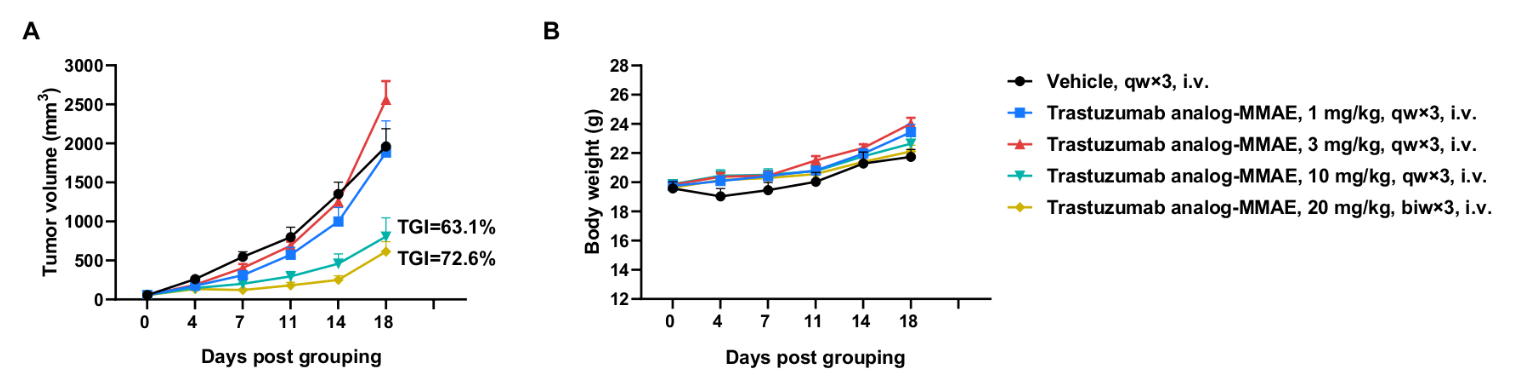
Antitumor activity of anti-human HER2 ADC (Trastuzumab analog-MMAE, in-house) in BALB/c mice. (A) Anti-human HER2 ADC inhibited B-hHER2 CT26.WT tumor growth in BALB/c mice. Murine colon cancer B-hHER2 CT26.WT cells were subcutaneously implanted into BALB/c mice (female, 9-week-old, n=6). Mice were grouped when tumor volume reached approximately 50 mm3, at which time they were intraperitoneally injected with anti-human HER2 ADC Trastuzumab analog-MMAE (in-house) indicated in panel. (B) Body weight changes during treatment. As shown in panel A, anti-human HER2 ADC was efficacious in controlling tumor growth in BALB/c mice, demonstrating that B-hHER2 CT26.WT provide a powerful preclinical model for in vivo evaluation of anti-human HER2 antibodies. Values are expressed as mean ± SEM.
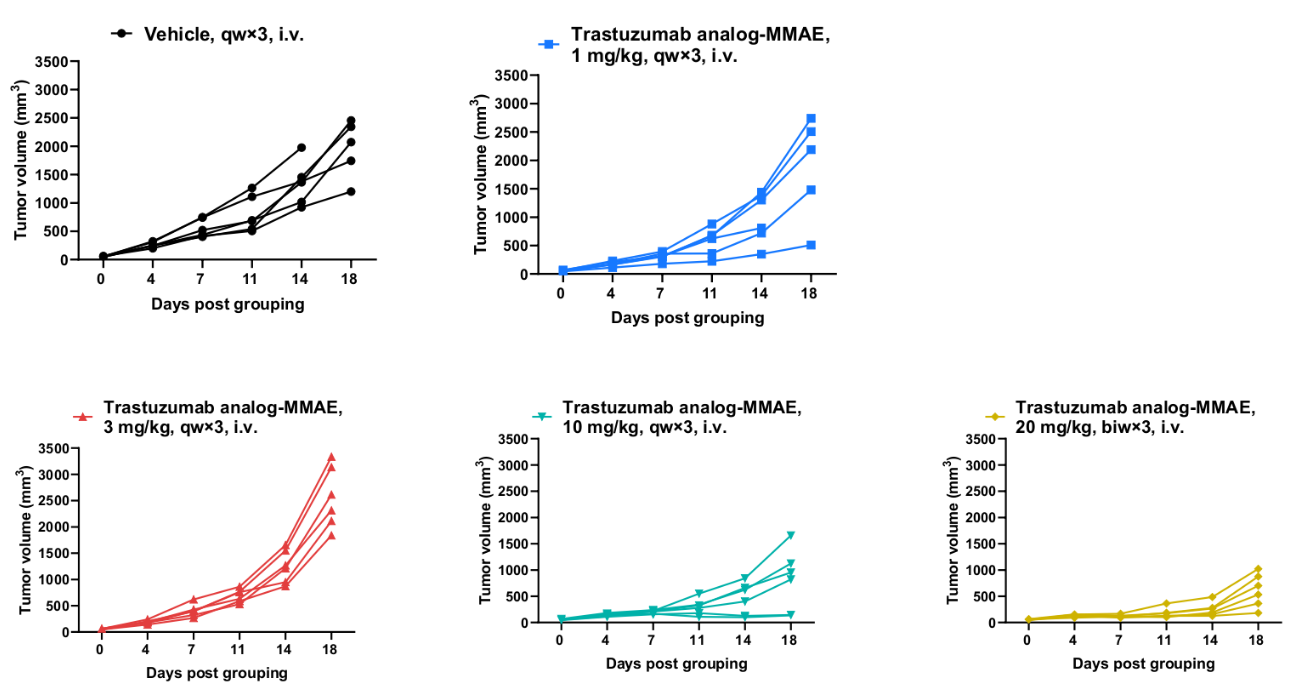
Antitumor activity of anti-human HER2 ADC (Trastuzumab analog-MMAE, in-house) in BALB/c mice. Murine colon cancer B-hHER2 CT26.WT cells were subcutaneously implanted into BALB/c mice (female, 9-week-old, n=6). Mice were grouped when tumor volume reached approximately 50 mm3, at which time they were intraperitoneally injected with anti-human HER2 ADC Trastuzumab analog-MMAE (in-house) indicated in panel.
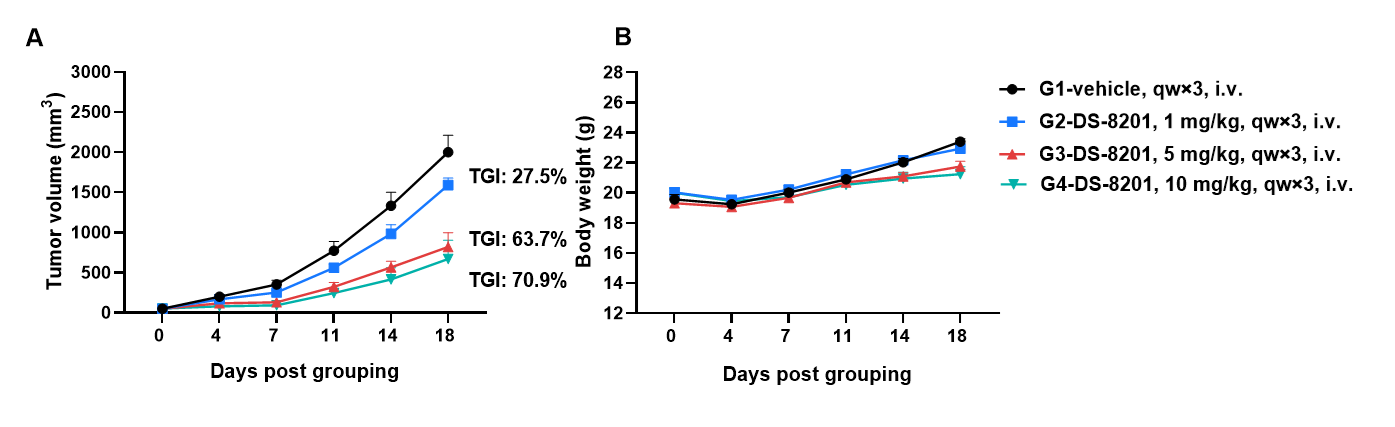
Anti tumor activity of anti-human HER2 antibody (DS8201, purchased from Daiichi Sankyo) in BALB/c mice. (A) Anti-human HER2 antibody inhibited B-hHER2 CT26.WT tumor growth in BALB/c mice. Murine colon cancer B-hHER2 CT26.WT cells were subcutaneously implanted into BALB/c mice (female, 8-week-old, n=6). Mice were grouped when tumor volume reached approximately 50 mm3, at which time they were intravenously injected with anti-human HER2 ADC DS-8201 (purchased from AstraZeneca/Daiichi Sankyo) indicated in panel. (B) Body weight changes during treatment. As shown in panel A, anti-human HER2 ADC DS-8201 (purchased from AstraZeneca/Daiichi Sankyo) was efficacious in controlling tumor growth in BALB/c mice in a dose-dependent manner, demonstrating that the BALB/C mice provide a powerful preclinical model for in vivo evaluation of anti-human HER2 antibodies. Values are expressed as mean ± SEM.
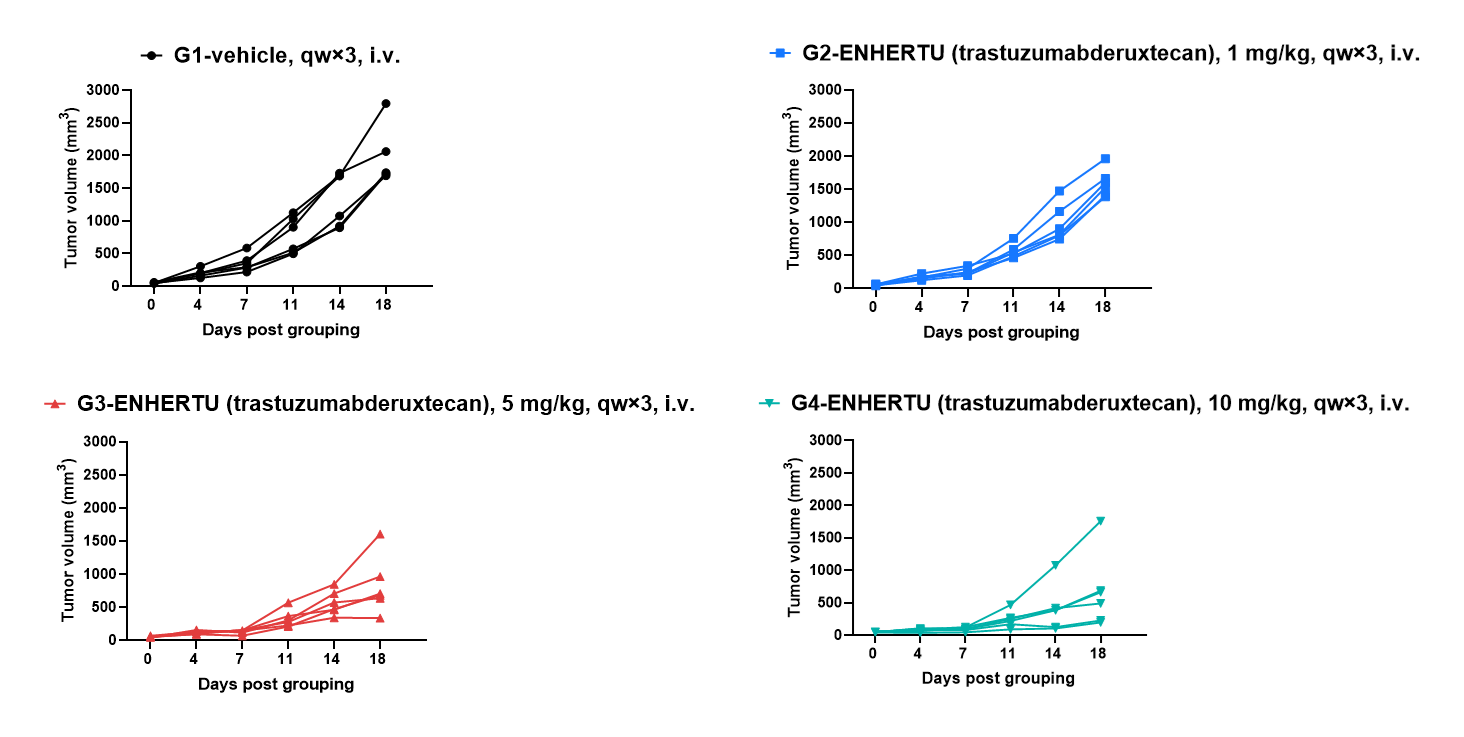
Antitumor activity of anti-human HER2 antibody (DS-8201, purchased from) in BALB/c mice. (A) Anti-human HER2 antibody inhibited B-hHER2 CT26.WT tumor growth in BALB/c mice. Murine colon cancer B-hHER2 CT26.WT cells were subcutaneously implanted into homozygous BALB/c mice (female, 8-week-old, n=6). Mice were grouped when tumor volume reached approximately 50 mm3, at which time they were intravenously injected with anti-human HER2 ADC DS-8201 (purchased from AstraZeneca/Daiichi Sankyo) indicated in panel.
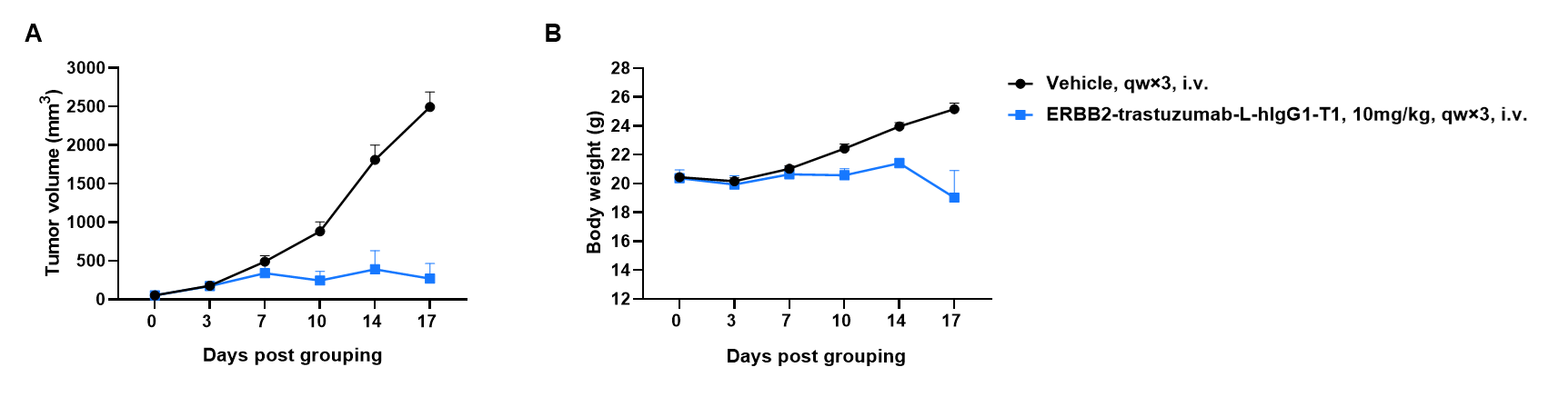
Antitumor activity of anti-human HER2 ADC (ERBB2-trastuzumab-L-hIgG1-T1, in-house) in B-hHER2 mice(C). (A) Anti-human HER2 ADC inhibited B-hHER2 CT26.WT tumor growth in B-hHER2 mice(C). Murine colon cancer B-hHER2 CT26.WT cells were subcutaneously implanted into B-hHER2 mice(C) (female, 8-week-old, n=6). Mice were grouped when tumor volume reached approximately 80 mm3, at which time they were intraperitoneally injected with anti-human HER2 ADC ERBB2-trastuzumab-L-hIgG1-T1 (in-house) indicated in panel. (B) Body weight changes during treatment. As shown in panel A, anti-human HER2 ADC was efficacious in controlling tumor growth in B-hHER2 mice(C), demonstrating that B-hHER2 CT26.WT provide a powerful preclinical model for in vivo evaluation of anti-human HER2 antibodies. Values are expressed as mean ± SEM.
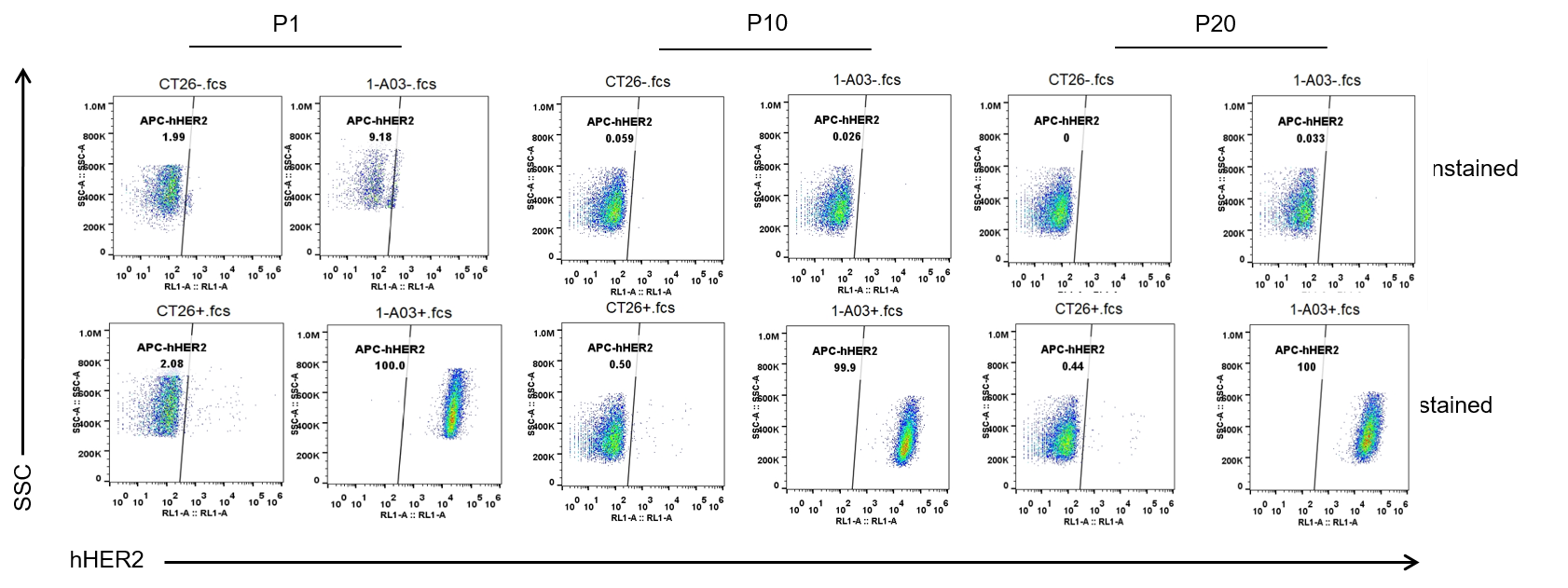
Passage stability analysis in B-hHER2 CT26.WT cells by flow cytometry. Single cell suspensions from wild-type CT26.WT and B-hHER2 CT26.WT cultures were stained with species-specific anti-HER2 antibody. Human HER2 was specifically detected on the surface of B-hHER2 CT26.WT cells and remained stable across multiple passages, with no significant change in expression levels observed. No expression was detected in wild-type CT26.WT cells.
The passage number for this cell line is calculated starting from the first subculture after revival from the master cell bank, which is designated as P1.