
CB17/lcr-PrkdcscidFcgr4tm2(FCGR3A)Bcgen/Bcgen • 111174
Gene targeting strategy for B-hCD16A mice(CB-17 SCID).
The regulatory region and exons 1~5 of mouse Fcgr4 gene that encode the full-length protein were replaced by human FCGR3A(F158V) exons 1~6 and regulatory region in B-hCD16A mice(CB-17 SCID).
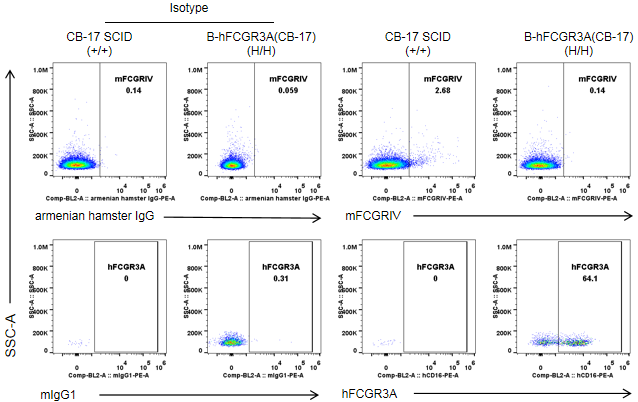
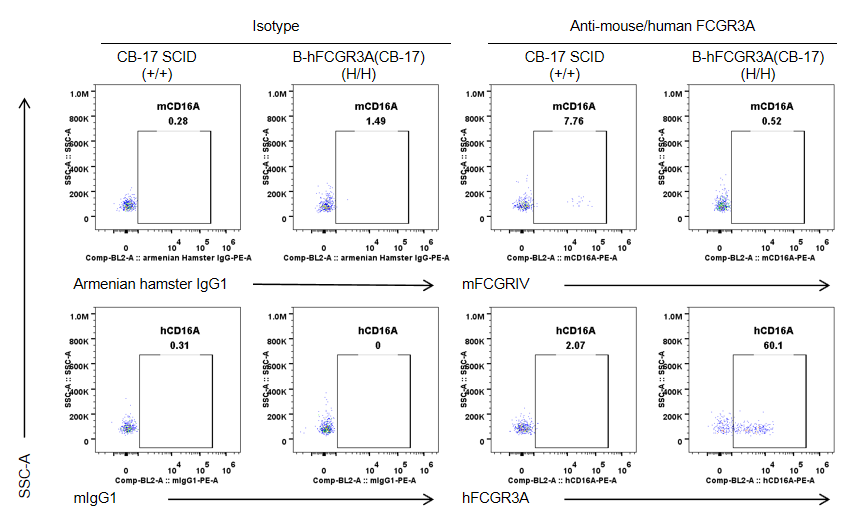
Strain specific FCGR3A expression analysis in wild-type CB-17 SCID mice and homozygous B-hFCGR3A (CB-17 SCID) by flow cytometry. Splenocytes were collected from wild-type CB-17 SCID mice (+/+) and homozygous B-hFCGR3A mice (CB-17 SCID) (H/H). Mouse FCGRIV was not detectable in NK cells of wild-type CB-17 SCID mice. Human FCGR3A was detectable in NK cells of B-hFCGR3A mice (CB-17 SCID). Note: the clone of anti-human CD16A antibody is 3G8.
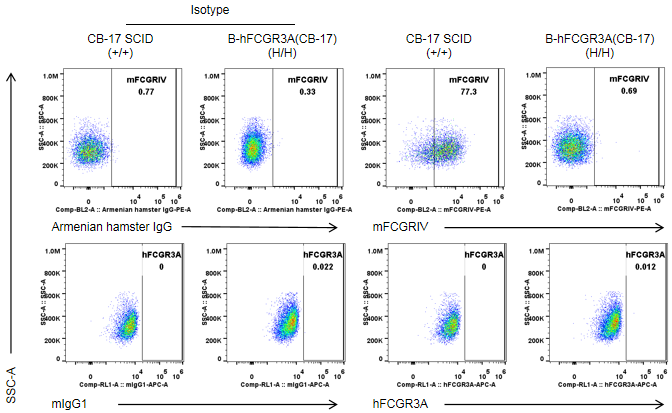
Strain specific FCGR3A expression analysis in wild-type CB-17 SCID mice and homozygous B-hFCGR3A(CB-17 SCID) by flow cytometry. Splenocytes were collected from wild-type CB-17 SCID mice(+/+) and homozygous B-hFCGR3A mice (CB-17 SCID) (H/H). Mouse FCGR3A was exclusively detectable in granulocytes of wild-type CB-17 SCID. Human FCGR3A was not detectable in granulocytes of wild-type CB-17 SCID mice and homozygous B-hFCGR3A mice (CB-17 SCID). Note: the clone of anti-human CD16A antibody is 3G8.
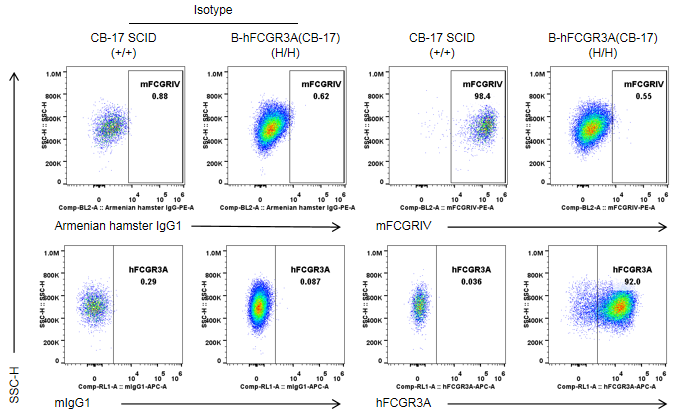
Strain specific FCGR3A expression analysis in wild-type CB-17 SCID mice and homozygous B-hFCGR3A(CB-17 SCID) by flow cytometry. Peritoneal exudative macrophages(PEMs) were collected from wild-type CB-17 SCID mice(+/+) and homozygous B-hFCGR3A mice (CB-17 SCID) (H/H). Mouse FCGRIV was exclusively detectable in PEMs of wild-type C57BL/6 mice. Human FCGR3A was exclusively detectable in PEMs of homozygous B-hFCGR3A mice (CB-17 SCID). Note: the clone of anti-human CD16A antibody is 3G8.
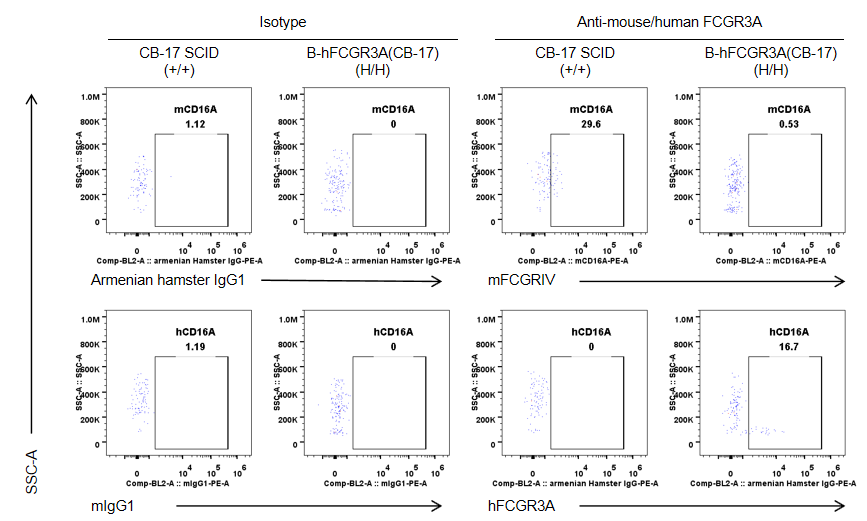
Strain specific CD16A expression analysis in wild-type CB-17 SCID mice and homozygous B-hCD16A (CB-17 SCID) by flow cytometry. Splenocytes were collected from wild-type CB-17 SCID mice(+/+) and homozygous B-hCD16A mice(CB-17 SCID) (H/H). Mouse CD16A was exclusively detectable in monocytes of wild-type CB-17 SCID mice. Human CD16A was exclusively detectable in monocytes of B-hCD16A mice(CB-17 SCID). Note: the clone of anti-human CD16A antibody is 3G8.
Strain specific CD16A expression analysis in wild-type CB-17 SCID mice and homozygous B-hCD16A (CB-17 SCID) by flow cytometry. Blood cells were collected from wild-type CB-17 SCID mice(+/+) and homozygous B-hCD16A mice(CB-17 SCID) (H/H). Mouse CD16A was exclusively detectable in monocytes of wild-type CB-17 SCID mice. Human CD16A was exclusively detectable in monocytes of B-hCD16A mice(CB-17 SCID). Note: the clone of anti-human CD16A antibody is 3G8.
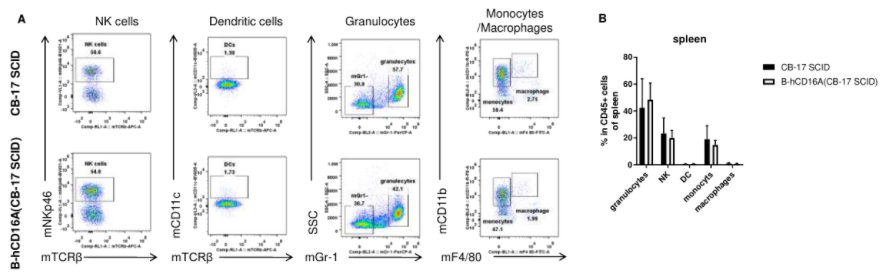
Analysis of spleen leukocyte subpopulations by FACS. Splenocytes were isolated from female wild-type CB-17 SCID and B-hCD16A mice(CB-17 SCID) (n=3, 8-week-old). Flow cytometry analysis of the splenocytes was performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live cells were gated for the CD45+ population and used for further analysis as indicated here. B. Results of FACS analysis. Percent of NK cells, dendritic cells, granulocytes, monocytes and macrophages in homozygous B-hCD16A mice(CB-17 SCID) were similar to those in the CB-17 SCID mice, demonstrating that CD16A humanized does not change the overall development, differentiation or distribution of these cell types in spleen. Values are expressed as mean ± SEM.
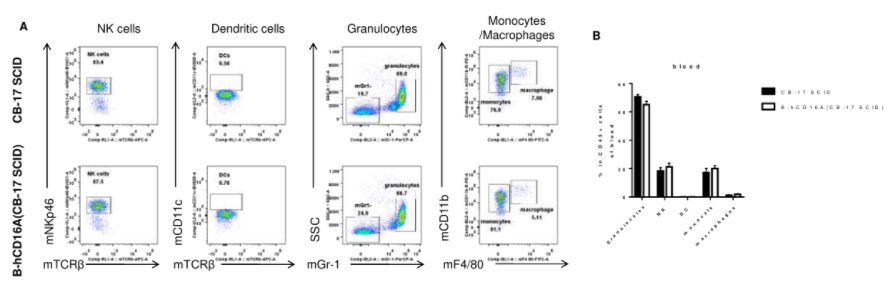
Analysis of blood leukocyte subpopulations by FACS. Blood were isolated from female wild-type CB-17 SCID and B-hCD16A mice(CB-17 SCID) (n=3, 8-week-old). Flow cytometry analysis of the blood leukocytes was performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live cells were gated for the CD45+ population and used for further analysis as indicated here. B. Results of FACS analysis. Percent of NK cells, dendritic cells, granulocytes, monocytes and macrophages in homozygous B-hCD16A mice(CB-17 SCID) were similar to those in the CB-17 SCID mice, demonstrating that CD16A humanized does not change the overall development, differentiation or distribution of these cell types in blood. Values are expressed as mean ± SEM.
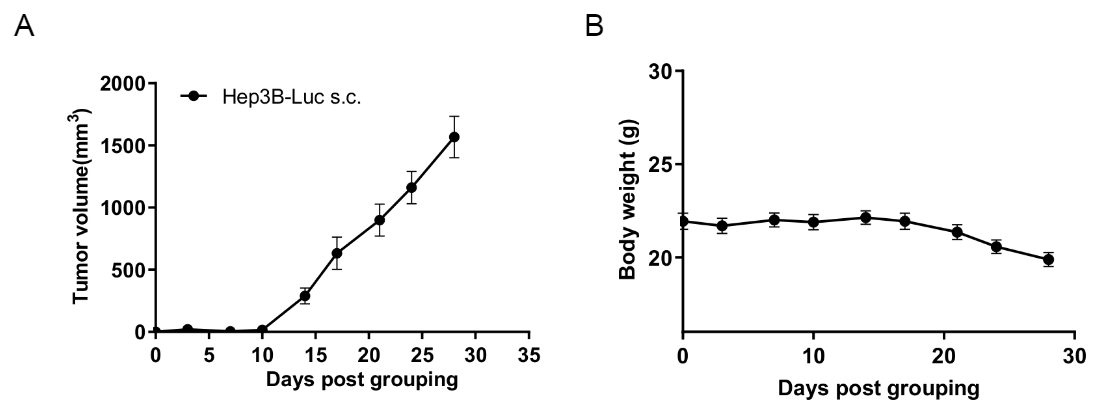
Subcutaneous CDX tumor growth of B-hHep3B-Luc cells in B-hCD16A mice(CB-17 SCID).
B-hHep3B-Luc cells(8x106) were subcutaneously implanted into B-hCD16A mice(CB-17 SCID) (female, 8-week-old, n=6). Tumor volume and body weight were measured twice a week. (A) Average tumor volume ± SEM. (B) Body weight (Mean± SEM). Volume was expressed in mm3 using the formula: V=0.5 × long diameter × short diameter2. As shown in panel A, B-hHep3B-Luc cells were able to establish tumors in B-hCD16A mice(CB-17 SCID) and can be used for efficacy studies.
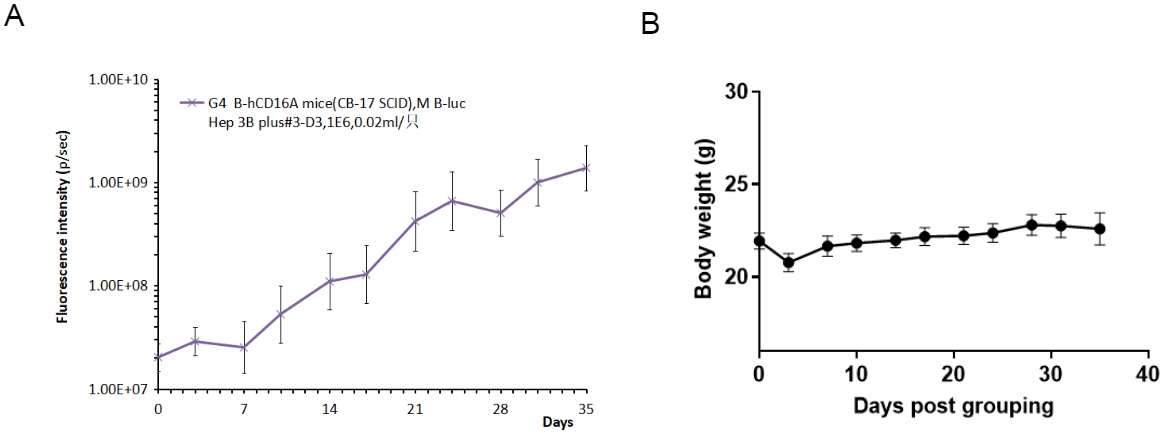
In situ CDX tumor growth of B-hHep3B-Luc cells in B-hCD16A mice (CB-17 SCID).
B-hHep3B-Luc cells (8x106) were in situ implanted into B-hCD16A mice (CB-17 SCID) (female, 8-week-old, n=6). Tumor fluorescence intensity and body weight were measured twice a week. (A) Average tumor volume ± SEM. (B) Body weight (Mean± SEM). As shown in panel A, B-hHep3B-Luc cells were able to establish tumors in B-hCD16A mice (CB-17 SCID) and can be used for efficacy studies.
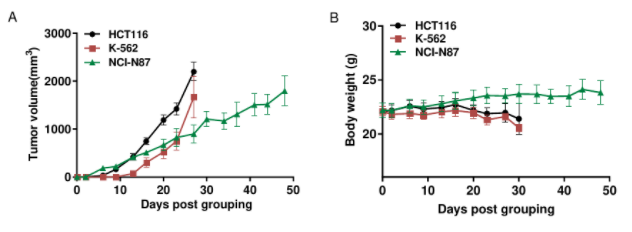
Subcutaneous CDX tumor growth in B-hCD16A mice(CB-17 SCID).
HC116 (5x106), K-562 (1x106) and NCI-N87 (1x107) were subcutaneously implanted into B-hCD16A mice(CB-17 SCID) (female, 8-week-old, n=6). Tumor volume and body weight were measured twice a week. (A) Average tumor volume ± SEM. (B) Body weight (Mean± SEM). Volume was expressed in mm3 using the formula: V=0.5 × long diameter × short diameter2. As shown in panel A, HCT116, K-562 and NCI-N87 were able to establish tumors in B-hCD16A mice(CB-17 SCID) and can be used for efficacy studies.
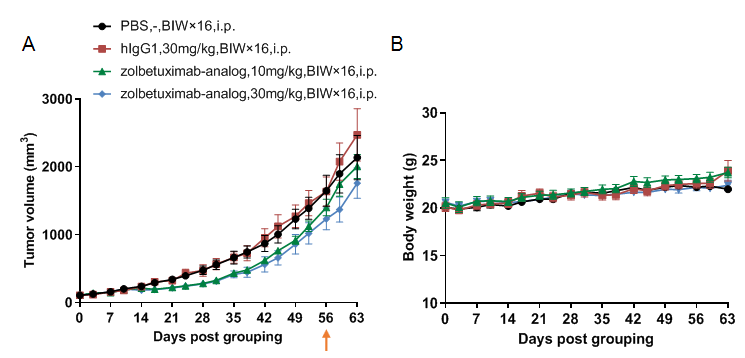
Antitumor activity of anti-human CLDN18.2 antibody in B-hCD16A mice (CB-17 SCID) bearing B-hCLDN18.2 BxPC-3 cells. (A) Anti-hCLDN18.2 antibody inhibited tumor growth. Human pancreas B-hCLDN18.2 BxPC-3 cells were subcutaneously implanted into homozygous B-hCD16A mice (CB-17 SCID) (female, 10 week-old, n=6). Mice were grouped when tumor volume reached approximately 100 mm3, at which time they were treated with antibodies in panel A. (B) Body weight changes during treatment. As shown in panel, B-hCD16A mice (CB-17 SCID) bearing B-hCLDN18.2 BxPC-3 cells provide a powerful preclinical model for in vivo evaluation of anti-CLDN18.2 antibodies. Values are expressed as mean ± SEM.
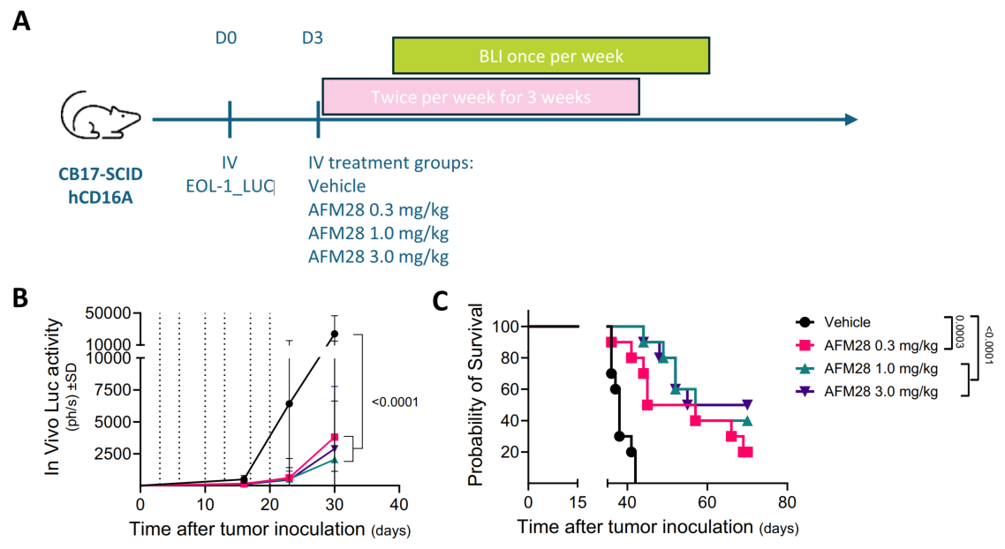
In vivo anti-leukemic activity of AFM28 against a human AML cell line.
A. Experimental timeline of the xenograft model using B-hCD16A mice (CB-17 SCID). On day 0, all mice (n = 40, 10 per group) were subjected to 1.2 Gy irradiation, followed by intravenous inoculation with 1 × 10⁶ EOL-1 Luc cells four hours later. Three days post-tumor implantation, the mice were grouped based on body weight and administered either vehicle or AFM28 at doses of 0.3, 1, or 3 mg/kg twice weekly for three weeks. Bioluminescence imaging (BLI) was conducted weekly beginning on day 15. Dashed lines indicate days of treatment administration.
B. Tumor growth, as measured by in vivo luciferase activity (expressed in photons per second, linear scale), is presented over the treatment period. Data are shown as mean ± SD (n = 10 mice/group). Statistical analysis was performed using two-way ANOVA followed by Tukey’s multiple comparisons test at day 30; corresponding p-values are indicated.
C. Survival curves were generated using the Kaplan–Meier method. Group comparisons were assessed by the log-rank (Mantel–Cox) test, with p-values shown in the figure.